- Y Diweddaraf sydd Ar Gael (Diwygiedig)
- Pwynt Penodol mewn Amser (31/12/2020)
- Gwreiddiol (Fel y’i mabwysiadwyd gan yr UE)
Commission Regulation (EC) No 2870/2000Dangos y teitl llawn
Commission Regulation (EC) No 2870/2000 of 19 December 2000 laying down Community reference methods for the analysis of spirits drinks
You are here:
- Rheoliadau yn deillio o’r UE
- 2000 No. 2870
- Annexes only
- Dangos Graddfa Ddaearyddol(e.e. Lloegr, Cymru, Yr Alban aca Gogledd Iwerddon)
- Dangos Llinell Amser Newidiadau
Rhagor o Adnoddau
PDF o Fersiynau Diwygiedig
- ddiwygiedig 26/04/20161.02 MB
- ddiwygiedig 04/12/20020.21 MB
When the UK left the EU, legislation.gov.uk published EU legislation that had been published by the EU up to IP completion day (31 December 2020 11.00 p.m.). On legislation.gov.uk, these items of legislation are kept up-to-date with any amendments made by the UK since then.
Mae hon yn eitem o ddeddfwriaeth sy’n deillio o’r UE
Mae unrhyw newidiadau sydd wedi cael eu gwneud yn barod gan y tîm yn ymddangos yn y cynnwys a chyfeirir atynt gydag anodiadau.Ar ôl y diwrnod ymadael bydd tair fersiwn o’r ddeddfwriaeth yma i’w gwirio at ddibenion gwahanol. Y fersiwn legislation.gov.uk yw’r fersiwn sy’n weithredol yn y Deyrnas Unedig. Y Fersiwn UE sydd ar EUR-lex ar hyn o bryd yw’r fersiwn sy’n weithredol yn yr UE h.y. efallai y bydd arnoch angen y fersiwn hon os byddwch yn gweithredu busnes yn yr UE. EUR-Lex Y fersiwn yn yr archif ar y we yw’r fersiwn swyddogol o’r ddeddfwriaeth fel yr oedd ar y diwrnod ymadael cyn cael ei chyhoeddi ar legislation.gov.uk ac unrhyw newidiadau ac effeithiau a weithredwyd yn y Deyrnas Unedig wedyn. Mae’r archif ar y we hefyd yn cynnwys cyfraith achos a ffurfiau mewn ieithoedd eraill o EUR-Lex. The EU Exit Web Archive legislation_originated_from_EU_p3
Changes over time for: Commission Regulation (EC) No 2870/2000 (Annexes only)
Alternative versions:
Status:
Point in time view as at 31/12/2020.
Changes to legislation:
There are currently no known outstanding effects for the Commission Regulation (EC) No 2870/2000.![]()
Changes to Legislation
Revised legislation carried on this site may not be fully up to date. At the current time any known changes or effects made by subsequent legislation have been applied to the text of the legislation you are viewing by the editorial team. Please see ‘Frequently Asked Questions’ for details regarding the timescales for which new effects are identified and recorded on this site.
ANNEXU.K.
I.DETERMINATION OF ALCOHOLIC STRENGTH BY VOLUME OF SPIRIT DRINKSU.K.
IntroductionU.K.
The reference method includes two Appendices:
:
Preparation of distillate
:
Measurement of density of distillate
1.ScopeU.K.
The method is suitable for the determination of the real alcoholic strength by volume of spirit drinks.
2.Normative ReferencesU.K.
ISO 3696:1987: Water for analytical laboratory use — Specifications and test methods.
3.Terms and DefinitionsU.K.
3.1. Reference temperature: U.K.
The reference temperature for the determination of alcoholic strength by volume, density and specific gravity of spirit drinks is 20 °C.
Note 1:U.K.
The term ‘at t °C’ is reserved for all determinations (of density or alcoholic strength by volume) expressed at a temperature other than the reference temperature of 20 °C.U.K.
3.2. Density: U.K.
The density is the mass per unit volume in vacuo of spirit drinks at 20 °C. It is expressed in kilograms per cubic metre and its symbol is ρ20 °C or ρ20.
3.3. Specific gravity: U.K.
The specific gravity is the ratio, expressed as a decimal number, of the density of spirit drinks at 20 °C to the density of water at the same temperature. It is denoted by the symbol d20 °C/20 °C or d20/20, or simply d when there is no possibility of confusion. The characteristic that was measured must be specified on the assay certificate using the above-defined symbols only.
Note 2:U.K.
It is possible to obtain the specific gravity from the density ρ20 at 20 °C: 

3.4. Real alcoholic strength by volume: U.K.
The real alcoholic strength by volume of spirit drinks is equal to the number of litres of ethyl alcohol contained in 100 l of a water-alcohol mixture having the same density as the alcohol or spirit after distillation. The reference values for alcoholic strength by volume (% vol) at 20 °C versus density at 20 °C for different water-alcohol mixtures that are to be used are those given in the international table adopted by the International Legal Metrology Organisation in its Recommendation No 22.
The general equation relating the alcoholic strength by volume and density of a water-alcohol mixture at a given temperature is given on page 40 in Chapter 3 ‘Alcoholic strength by volume’ of the Annex to Commission Regulation (EEC) No 2676/90 (OJ L 272, 3.10.1990, p. 1) or in the manual of analysis methods of the OIV (1994) (p. 17).
Note 3:U.K.
For liqueurs and crèmes for which it is very difficult to measure volume accurately the sample must be weighed and the alcoholic strength is calculated first by mass.U.K.
where
=
alcoholic strength by mass,
=
789,24 kg/m3
4.PrincipleU.K.
Following distillation, the alcoholic strength by volume of the distillate is determined by pycnometry, electronic densimetry, or densimetry using a hydrostatic balance.
APPENDIX I: PREPARATION OF DISTILLATEU.K.
1.ScopeU.K.
The method is suitable for the preparation of distillates to be used to determine the real alcoholic strength by volume of spirit drinks.
2.PrincipleU.K.
The spirits are distilled to separate the ethyl alcohol and other volatile compounds from the extractive matter (substances which do not distil).
3.Reagents and MaterialsU.K.
3.1.Anti-bumping granules.U.K.
3.2.Concentrated antifoam emulsion (for crème liqueurs).U.K.
4.Apparatus and equipmentU.K.
Usual laboratory apparatus and in particular the following.
Water bath capable of being maintained at 10 °C to 15 °C.
Water bath capable of being maintained at 20 °C (± 0,2 °C).
Class A volumetric flasks, 100 ml and 200 ml, that have been certified to 0,1 % and 0,15 % respectively.
Distillation apparatus:
General requirementsU.K.
The distillation apparatus must meet the following specifications:
the number of joints must be no more than the strict minimum needed to ensure the system is leak-tight,
inclusion of a device designed to prevent priming (entrainment of the boiling liquid by the vapour) and to regularise the distillation rate of alcohol-rich vapours,
rapid and complete condensation of the alcohol vapours,
collection of the first distillation fractions in an aqueous medium.
The heat source must be used with a suitable heat-diffuser to prevent any pyrogenic reaction involving the extractive matter.
An example of a suitable distillation apparatus is shown in Figure 1 and includes the following parts:
round-bottomed flask, 1 litre, with a standardised ground-glass joint,
rectifying column at least 20-cm high (a Vigreux column, for example),
elbow connector with an approximately 10-cm-long straight-rimmed condenser (a West-type condenser) fitted vertically,
cooling coil, 40-cm long,
drawn-out tube, taking the distillate to the bottom of a graduated collecting flask containing a small amount of water.
Note:U.K.
The apparatus described above is intended for a sample of least 200 ml. However, a smaller sample size (100 ml) can be distilled by using a smaller distillation flask, provided a splashhead or some other device to prevent entrainment is used.U.K.
5.Storage of test samplesU.K.
Samples are stored at room temperature prior to analysis.
6.ProcedureU.K.
Preliminary remark:
Distillation may also be by the procedure published by IUPAC (1968).
6.1.Distillation apparatus verification.U.K.
The apparatus used must be capable of the following:
The distillation of 200 ml of a water-alcohol solution with known concentration close to 50 % vol must not cause a loss of alcohol of more than 0,1 % vol.
6.2.Spirit drinks with alcoholic strength below 50 % vol.U.K.
Measure out 200 ml of the spirit into a volumetric flask.
Record the temperature of this liquid, or maintain at standard temperature (20 °C).
Pour the sample into the round-bottomed flask of the distillation apparatus and rinse the volumetric flask with three aliquots each of approximately 20 ml of distilled water. Add each rinse water aliquot to the contents of the distillation flask.
Note:U.K.
This 60-ml dilution is sufficient for spirits containing less than 250 g of dry extract per litre. Otherwise, to prevent pyrolysis, the volume of rinse water must be at least 70 ml if the dry extract concentration is 300 g/l, 85 ml for 400 g/l dry extract, and 100 ml for 500 g/l dry extract (some fruit liqueurs or crèmes). Adjust these volumes proportionally for different sample volumes.U.K.
Add a few anti-bumping granules (3.1) (and antifoam for crème liqueurs).
Pour 20 ml of distilled water into the original 200 ml volumetric flask that will be used to hold the distillate. This flask must then be placed in a cold water bath (4.1) (10 to 15 °C for aniseed-flavoured spirit drinks).
Distil, avoiding entrainment and charring, occasionally agitating the contents of the flask, until the level of distillate is a few millimetres below the calibration mark of the volumetric flask.
When the temperature of this distillate has been brought down to within 0,5 °C of the liquid's initial temperature, make up to the mark with distilled water and mix thoroughly.
This distillate is used for the determination of alcoholic strength by volume (Appendix II)
6.3.Spirit drinks with alcoholic strength above 50 % vol.U.K.
Measure out 100 ml of the spirit drink into a 100-ml volumetric flask and pour into the round bottomed flask of the distillation apparatus.
Rinse the volumetric flask several times with distilled water and add the washings to the contents of the round-bottomed distillation flask. Use enough water to bring the flask's contents up to approximately 230 ml.
Pour 20 ml of distilled water into a 200-ml volumetric flask that will be used to hold the distillate. This flask must then be placed in a cold water bath (4.1) (10 to 15 °C for aniseed-flavoured spirits).
Distil, agitating the contents occasionally, until the level of distillate is a few millimetres below the calibration mark of the 200-ml volumetric flask.
When the temperature of this distillate has been brought down to within 0,5 °C of the liquid's initial temperature, make up to the mark with distilled water and mix thoroughly.
This distillate is used for the determination of alcoholic strength by volume (Appendix II)
Note:U.K.
The alcoholic strength by volume of the spirit drink is twice the alcoholic strength of the distillate.U.K.
APPENDIX II: MEASUREMENT OF DENSITY OF DISTILLATEU.K.
METHOD A:DETERMINATION OF REAL ALCOHOLIC STRENGTH BY VOLUME OF SPIRIT DRINKS — MEASUREMENT BY PYCNOMETRYU.K.
A.1.PrincipleU.K.
The alcoholic strength by volume is obtained from the density of the distillate measured by pycnometry.
A.2.Reagents and materialsU.K.
During the analysis, unless otherwise is stated, use only reagents of recognised analytical grade and water of at least grade 3 as defined in ISO 3696:1987.
A.2.1.Sodium chloride solution (2 % w/v)U.K.
To prepare 1 litre, weigh out 20 g of sodium chloride and dissolve to 1 litre using water.
A.3.Apparatus and EquipmentU.K.
Usual laboratory apparatus and in particular the following:
Analytical balance capable of reading 0,1 mg.
Thermometer, with ground glass joint, calibrated in tenths of a degree from 10 to 30 °C. This thermometer must be certified or checked against a certified thermometer.
Pyrex-glass pycnometer of approximately 100 ml capacity fitted with a removable ground-glass thermometer (A.3.2). The pycnometer has a side tube 25 mm in length and 1 mm (maximum) in internal diameter ending in a conical ground joint. Other pycnometers as described in ISO 3507, e.g. 50 ml may be used if appropriate.
A tare bottle of the same external volume (to within 1 ml) as the pycnometer and with a mass equal to the mass of the pycnometer filled with a liquid of density 1,01 (sodium chloride solution A.2.1).
Thermally insulated jacket that fits the body of the pycnometer exactly.
Note 1:U.K.
The method for determining the densities in vacuo of spirits calls for the use of a twin-pan balance, a pycnometer and a tare bottle of the same outside external volume to cancel out the effect of air buoyancy at any given moment. This simple technique may be applied using a single-pan balance provided that the tare bottle is weighed again to monitor changes in air buoyancy over time.U.K.
A.4.ProcedureU.K.
Preliminary remarks:
The following procedure is described for the use of 100-ml pycnometer for determination of the alcoholic strength; this gives the best accuracy. However, it is also possible to use a smaller pycnometer, for example 50 ml.
A.4.1.Calibration of pycnometerU.K.
The pycnometer is calibrated by determining the following parameters:
tare of the empty pycnometer,
volume of the pycnometer at 20 °C,
mass of the water-filled pycnometer at 20 °C.
A.4.1.1.Calibration using a single-pan balance:U.K.
Determine:
the mass of the clean, dry pycnometer (P),
the mass of the water-filled pycnometer at t °C (P1),
the mass of the tare bottle (T0).
A.4.1.1.1.Weigh the clean, dry pycnometer (P).U.K.
A.4.1.1.2.Fill the pycnometer carefully with distilled water at ambient temperature and fit the thermometer.U.K.
Carefully wipe the pycnometer dry and place it in the thermally-insulated jacket. Agitate by inverting the container until the thermometer's temperature reading is constant.
Set the pycnometer flush with the upper rim of the side tube. Read the temperature t °C carefully and if necessary correct for any inaccuracies in the temperature scale.
Weigh the water-filled pycnometer (P1).
A.4.1.1.3.Weigh the tare bottle (T0).U.K.
A.4.1.1.4.CalculationU.K.
Tare of the empty pycnometer = P – m
where m is the mass of air in the pycnometer.
m = 0,0012 × (P1 – P)
Note 2:U.K.
0,0012 is the density of dry air at 20 °C at a pressure of 760 mm HgU.K.
Volume of the pycnometer at 20 °C:
where Ft is the factor for temperature t °C taken from Table I of Chapter 1 ‘Density and specific gravity’ of the Annex to Regulation (EEC) No 2676/90 (p. 10).
V20 °C must be known to the nearest 0,001 ml.
Mass of water in the pycnometer at 20 °C:
where 0,998203 is the density of water at 20 °C.
Note 3:U.K.
If necessary, the value 0,99715 of the density in air can be used and the alcoholic strength calculated with reference to the corresponding density in HM Customs and Excise tables in air.U.K.
A.4.1.2.Calibration method using a twin-pan balance:U.K.
Place the tare bottle on the left-hand pan and the clean, dry pycnometer with its collecting stopper on the right-hand pan. Balance them by placing weights on the pycnometer side: p grams.
Fill the pycnometer carefully with distilled water at ambient temperature and fit the thermometer; carefully wipe the pycnometer dry and place it in the thermally insulated jacket; agitate by inverting the container until the thermometer's temperature reading is constant.
Accurately adjust the level to the upper rim of the side tube. Clean the side tube, fit the collecting stopper; read the temperature t °C carefully and if necessary correct for any inaccuracies in the temperature scale.
Weigh the water-filled pycnometer, with p′ the weight in grams making up the equilibrium.
CalculationU.K.
Tare of the empty pycnometer = p + m
where m is the mass of air in the pycnometer.
m = 0,0012 × (p – p′)
Volume of the pycnometer at 20 °C:
where Ft is the factor for temperature t °C taken from Table I of Chapter 1 ‘Density and specific gravity’ of the Annex to Regulation (EEC) No 2676/90 (p. 10).
V20 °C must be known to the nearest 0,001 ml.
Mass of water in the pycnometer at 20 °C:
where 0,998203 is the density of water at 20 °C.
A.4.2.Determination of alcoholic strength of test sampleU.K.
Using a single-pan balance.
Weigh the tare bottle, weight T1.
Weigh the pycnometer with the prepared distillate (see Appendix I), P2 is its weight at t °C.
CalculationU.K.
Mass of empty pycnometer at moment of measuring
= P - m + dT
Mass of the liquid in the pycnometer at t °C
= P2 - (P - m + dT)
Density at t °C in g/ml
Express the density at t °C in kilograms per m3 by multiplying ρt °C by 1 000, the value being known as ρt.
Correct ρt to 20 using the table of densities ρT for water-alcohol mixtures (Table II of Appendix II to the OIV's manual of analysis methods (1994), pp. 17-29).
In the table find the horizontal line corresponding to temperature T in whole degrees immediately below t °C, the smallest density above ρt. Use the table difference found below that density to calculate the density ρt of the spirit at that temperature T in whole degrees.
Using the whole temperature line, calculate the difference between density ρ′ in the table immediately above ρt and the calculated density ρt. Divide that difference by the table difference found to the right of density ρ′. The quotient provides the decimal portion of the alcoholic strength while the integer of the alcoholic strength is found at the top of the column in which density ρ′ is found (Dt, the alcoholic strength).
Note 4:U.K.
Alternatively keep the pycnometer in a water bath maintained at 20 °C (± 0,2 °C) when making up to the mark.U.K.
ResultU.K.
Using the density ρ20 calculate the real alcoholic strength using the alcoholic strength tables identified below:
The table giving the value of the alcoholic strength by volume (% vol) at 20 °C as a function of the density at 20 °C of water-alcohol mixtures is the international table adopted by the International Legal Metrology Organisation in its Recommendation No 22.
A.5.Method performance characteristics (precision)U.K.
A.5.1.Statistical results of the interlaboratory testU.K.
The following data were obtained from an international method performance study carried out to internationally agreed procedures [1] [2].
| Year of interlaboratory test | 1 997 |
| Number of laboratories | 20 |
| Number of samples | 6 |
METHOD B:DETERMINATION OF REAL ALCOHOLIC STRENGTH BY VOLUME OF SPIRIT DRINKS — MEASUREMENT BY ELECTRONIC DENSIMETRY (BASED ON THE RESONANT FREQUENCY OSCILLATION OF A SAMPLE IN AN OSCILLATION CELL)U.K.
B.1.PrincipleU.K.
The liquid's density is determined by electronic measurement of the oscillations of a vibrating U-tube. To perform this measurement, the sample is added to an oscillating system, whose specific oscillation frequency is thus modified by the added mass.
B.2.Reagents and materialsU.K.
During the analysis, unless otherwise is stated, use only reagents of recognised analytical grade and water of at least grade 3 as defined in ISO 3696:1987.
Acetone (CAS 666-52-4) or absolute alcohol
Dry air.
B.3.Apparatus and equipmentU.K.
Usual laboratory apparatus and in particular the following:
Digital display densimeterU.K.
Electronic densimeter for performing such measurements must be capable of expressing density in g/ml to 5 decimal places.
Note 1:U.K.
The densimeter should be placed on a perfectly stable stand that is insulated from all vibrations.U.K.
Temperature regulationU.K.
The densimeter's performance is valid only if the measuring cell is connected to a built-in temperature regulator that can achieve the same temperature stability of ± 0,02 °C or better.
Note 2:U.K.
The precise setting and monitoring of the temperature in the measuring cell are very important, for an error of 0,1 °C can lead to a variation in density of the order of 0,1 kg/m3.U.K.
Sample injection syringes or auto sampler.
B.4.ProcedureU.K.
Calibration of the densimeterU.K.
The apparatus must be calibrated according to the instrument manufacturer's instructions when it is first put into service. It must be recalibrated regularly and checked against a certified reference standard or an internal laboratory reference solution based on a certified reference standard.
Determination of sample densityU.K.
If required prior to measurement clean and dry the cell with acetone or absolute alcohol and dry air. Rinse the cell with the sample.
Inject the sample into the cell (using a syringe or autosampler) so that the cell is completely filled. During the filling operation make sure that all air bubbles are completely eliminated. The sample must be homogeneous and must not contain any solid particles. Any suspended matter should be removed by filtration prior to analysis.
Once the reading has stabilised, record the density ρ20 or the alcoholic strength displayed by the densimeter.
ResultU.K.
When the density ρ20 is used, calculate the real alcoholic strength using the alcoholic strength tables identified below:
The table giving the value of the alcoholic strength by volume (% vol) at 20 °C as a function of the density at 20 °C of water-alcohol mixtures is the international table adopted by the International Legal Metrology Organisation in its Recommendation No 22.
B.5.Method performance characteristics (precision)U.K.
B.5.1.Statistical results of the interlaboratory testU.K.
The following data were obtained from an international method performance study carried out to internationally agreed procedures [1] [2].
| Year of interlaboratory test | 1 997 |
| Number of laboratories | 16 |
| Number of samples | 6 |
METHOD C:DETERMINATION OF REAL ALCOHOLIC STRENGTH BY VOLUME OF SPIRIT DRINKS — MEASUREMENT BY DENSIMETRY USING HYDROSTATIC BALANCEU.K.
C.1.PrincipleU.K.
The alcoholic strength of spirits can be measured by densimetry using a hydrostatic balance based on Archimedes' principle according to which a body immersed in a liquid receives a vertical upward thrust from the liquid equal to the weight of liquid displaced.
C.2.Reagents and materialsU.K.
During the analysis, unless otherwise is stated, use only reagents of recognised analytical grade and water of at least grade 3 as defined in ISO 3696:1987.
C.2.1.Float cleaning solution (sodium hydroxide, 30 % w/v)U.K.
To prepare 100 ml, weigh 30 g of sodium hydroxide and make up to volume using 96 % volume ethanol.
C.3.Apparatus and EquipmentU.K.
Usual laboratory apparatus and in particular the following:
Single-pan hydrostatic balance with a sensitivity of 1 mg.
Float with a volume of at least 20 ml, specially adapted to the balance, suspended with a thread of diameter not exceeding 0,1 mm.
Measuring cylinder bearing a level mark. The float must be capable of being contained completely within the volume of the cylinder located below the mark; the surface of the liquid may only be penetrated by the supporting thread. The measuring cylinder must have an internal diameter at least 6 mm larger than that of the float.
Thermometer (or temperature-measuring probe) graduated in degrees and tenths of a degree from 10 to 40 °C, calibrated to 0,05 °C.
Weights, calibrated by a recognised certifying body.
Note 1:U.K.
Use of a twin-pan balance is also possible; the principle is described in Chapter 1 ‘Density and specific gravity’ of the Annex to Regulation (EEC) No 2676/90 (p. 7).U.K.
C.4.ProcedureU.K.
The float and measuring cylinder must be cleaned between each measurement with distilled water, dried with soft laboratory paper which does not shed fibres and rinsed with the solution whose density is to be determined. Measurements must be made as soon as the apparatus has reached stability so as to restrict alcohol loss by evaporation.
Calibration of the balanceU.K.
Although balances usually have an internal calibration system, the hydrostatic balance must be capable of calibration with weights checked by an official certifying body.
Calibration of the floatU.K.
Fill the measuring cylinder to the mark with double-distilled water (or water of equivalent purity, e.g. microfiltered water with a conductivity of 18,2 MΩ/cm) at a temperature between 15 and 25 °C but preferably at 20 °C.
Immerse the float and the thermometer, stir, read off the density of the liquid from the apparatus and, if necessary, correct the reading so that it is equal to that of the water at measurement temperature.
Control using a water-alcohol solutionU.K.
Fill the measuring cylinder to the mark with a water-alcohol mixture of known strength at a temperature between 15 and 25 °C but preferably at 20 °C.
Immerse the float and the thermometer, stir, read off the density of the liquid (or the alcoholic strength if this is possible) from the apparatus. The alcoholic strength thus established should be equal to the previously determined alcoholic strength.
Note 2:U.K.
This solution of known alcoholic strength can also be used to calibrate the float instead of double-distilled water.U.K.
Measurement of the density of a distillate (or of its alcoholic strength if the apparatus allows)U.K.
Pour the test sample into the measuring cylinder up to the graduation mark.
Immerse the float and the thermometer, stir, read off the density of the liquid (or the alcoholic strength if this is possible) from the apparatus. Note the temperature if the density is measured at t °C (ρt).
Correct ρt to 20 using the table of densities ρT for water-alcohol mixtures (Table II of Annex II to the OIV's Manual of analysis methods (1994), pp. 17-29).
Cleaning of float and measuring cylinderU.K.
Immerse the float in the float cleaning solution in the measuring cylinder.
Allow to soak for one hour spinning the float periodically.
Rinse with copious amounts of tap water followed by distilled water.
Dry with soft laboratory paper which does not shed fibres.
Carry out this procedure when the float is first used and then regularly as required.
ResultU.K.
Using the density ρ20 calculate the real alcoholic strength using the alcoholic strength tables identified below.
The table giving the value of the alcoholic strength by volume (% vol) at 20 °C as a function of the density at 20 °C of water-alcohol mixtures is the international table adopted by the International Legal Metrology Organisation in its Recommendation No 22.
C.5.Method performance characteristics (precision)U.K.
C.5.1.Statistical results of the interlaboratory testU.K.
The following data were obtained from an international method performance study carried out to internationally agreed procedures [1] [2].
| Year of interlaboratory test | 1 997 |
| Number of laboratories | 12 |
| Number of samples | 6 |
1-litre round-bottomed flask with standardised spherical ground-glass joint.
20-cm Vigreux rectifying column.
10-cm straight-rimmed West condenser.
40-cm cooling coil.
II.DETERMINATION OF TOTAL DRY EXTRACT OF SPIRIT DRINKS BY GRAVIMETRYU.K.
1.ScopeU.K.
Regulation (EEC) No 1576/89 provides for this method only for aquavit for which the dry extract is limited to 15 g/l.
2.Normative ReferencesU.K.
ISO 3696:1987: Water for analytical laboratory use — Specifications and test methods.
3.DefinitionU.K.
The total dry extract or total dry matter includes all matter that is non-volatile under specified physical conditions.
4.PrincipleU.K.
Weighing of the residue left by evaporation of the spirit on a boiling water bath and drying in a drying oven.
5.Apparatus and EquipmentU.K.
Flat-bottomed cylindrical evaporating dish 55 mm in diameter.
Boiling water bath.
25 ml pipette, class A.
Drying oven.
Desiccator.
Analytical balance accurate to 0,1 mg.
6.Sampling and samplesU.K.
Samples are stored at room temperature prior to analysis.
7.ProcedureU.K.
7.1.Pipette 25 ml of the spirit containing less than 15 g/l dry matter into a previously weighed flat-bottomed cylindrical evaporating dish 55 mm in diameter. During the first hour of evaporation the evaporating dish is placed on the lid of a boiling water bath so that the liquid will not boil, as this could lead to losses through splattering. Leave one more hour directly in contact with the steam of the boiling water bath.U.K.
7.2.Complete the drying by placing the evaporating dish in a drying oven at 105 °C ± 3 °C for two hours. Allow the evaporating dish to cool in a desiccator and weigh the evaporating dish and its contents.U.K.
8.CalculationU.K.
The mass of the residue multiplied by 40 is equal to the dry extract contained in the spirit and it must be expressed in g/l to one decimal place.
9.Method performance characteristics (precision)U.K.
9.1.Statistical results of the interlaboratory testU.K.
The following data were obtained from an international method performance study carried out to internationally agreed procedures [1] [2].
| Year of interlaboratory test | 1 997 |
| Number of laboratories | 10 |
| Number of samples | 4 |
III.DETERMINATION OF VOLATILE SUBSTANCES AND METHANOL OF SPIRIT DRINKSU.K.
III.1.GENERAL REMARKSU.K.
1.DefinitionsU.K.
[F1Regulation 110/2008] sets minimum levels of volatile compounds other than ethanol and methanol for a series of spirit drinks (rum, spirits of viticultural origin, fruit spirits, etc.). For this series of drinks only, these levels are conventionally considered to be equivalent to the sum of the concentrations of:
Textual Amendments
F1Words in Annex Ch. 3 Pt. 3.1 para. 1 substituted (31.12.2020) by The Agricultural Products, Food and Drink (Amendment etc.) (EU Exit) Regulations 2020 (S.I. 2020/1637), regs. 1(7), 16(6)(a)(i)
volatile acids expressed as acetic acid;
aldehydes expressed as ethanal by the sum of ethanal (acetaldehyde) and the ethanal fraction contained in 1,1-diethoxyethane (acetal);
the following higher alcohols: propan-1-ol, butan-1-ol, butan-2-ol, 2-methylpropan-1-ol, assayed by individual alcohol and 2-methylbutan-1-ol, and 3-methylbutan-1-ol assayed as individual alcohol or the sum of the two;
ethyl acetate.
The following are the conventional methods for measuring volatile compounds:
the volatile acids by means of volatile acidity,
the aldehydes (ethanal and acetal), ethyl acetate and the alcohols by means of gas chromatography (GPC).
2.Gas chromatographic analysis of volatile compoundsU.K.
Gas chromatographic assays of volatile compounds other than those set out above may prove particularly interesting as a means of determining both the origin of the raw material used in the distillation and the actual conditions of distillation.
Some spirits contain other volatile components, such as aromatic compounds, which are characteristic of the raw materials used to obtain the alcohol, of the aroma of the spirit drink and of the special features of the preparation of the spirit. These compounds are important for evaluating the requirements set out in [F2Regulation 110/2008].
Textual Amendments
F2Words in Annex Ch. 3 Pt. 3.1 para. 2 substituted (31.12.2020) by The Agricultural Products, Food and Drink (Amendment etc.) (EU Exit) Regulations 2020 (S.I. 2020/1637), regs. 1(7), 16(6)(a)(ii)
III.2.GAS CHROMATOGRAPHIC DETERMINATION OF VOLATILE CONGENERS: ALDEHYDES, HIGHER ALCOHOLS, ETHYL ACETATE AND METHANOLU.K.
1.ScopeU.K.
This method is suitable for use for the determination of 1,1-diethoxyethane (acetal), 2-methylbutan-1-ol (active amyl alcohol), 3-methylbutan-1-ol (isoamyl alcohol), methanol (methyl alcohol), ethyl ethanoate (ethyl acetate), butan-1-ol (n-butanol), butan-2-ol (sec-butanol), 2-methylpropan-1-ol (isobutyl alcohol), propan-1-ol (n-propanol) and ethanal (acetaldehyde) in spirit drinks using gas chromatography. The method uses an internal standard, for example pentan-3-ol. The concentrations of the analytes are expressed as grams per 100 litres of absolute alcohol; the alcoholic strength of the product must be determined prior to analysis. The spirit drinks that can be analysed using this method include whisky, brandy, rum, wine spirit, fruit spirit and grape marc spirit.
2.Normative ReferencesU.K.
ISO 3696:1987: Water for analytical laboratory use — Specifications and test methods.
3.DefinitionU.K.
Congeners are volatile substances formed along with ethanol during fermentation, distillation and maturation of spirit drinks.
4.PrincipleU.K.
Congeners in spirit drinks are determined by direct injection of the spirit drink, or appropriately diluted spirit drink, into a gas chromatography (GC) system. A suitable internal standard is added to the spirit drink prior to injection. The congeners are separated by temperature programming on a suitable column and are detected using a flame ionisation detector (FID). The concentration of each congener is determined with respect to the internal standard from response factors, which are obtained during calibration under the same chromatographic conditions as those of the spirit drink analysis.
5.Reagents and materialsU.K.
Unless otherwise stated, use only reagents of a purity greater than 97 %, purchased from an ISO-accredited supplier with a certificate of purity, free from other congeners at test dilution (this may be confirmed by injection of individual congener standards at the test dilution using GC conditions as in 6.4) and only water of at least grade 3 as defined in ISO 3696. Acetal and acetaldehyde must be stored in the dark at < 5 °C, all other reagents may be stored at room temperature.
Ethanol absolute (CAS 64-17-5).
Methanol (CAS 67-56-1).
Propan-1-ol (CAS 71-23-8).
2-methylpropan-1-ol (CAS 78-33-1).
Acceptable internal standards: pentan-3-ol (CAS 584-02-1), pentan-1-ol (CAS 71-41-0), 4-methylpentan-1-ol (CAS 626-89-1) or methyl nonanoate (CAS 1731-84-6).
2-methylbutan-1-ol (CAS 137-32-6).
3-methylbutan-1-ol (CAS 123-51-3).
Ethyl acetate (CAS 141-78-6).
Butan-1-ol (CAS 71-36-3).
Butan-2-ol (CAS 78-92-2).
Acetaldehyde (CAS 75-07-0).
Acetal (CAS 105-57-7).
40 % v/v ethanol solution
To prepare 400 ml/l ethanol solution pour 400 ml ethanol (5.1) into a 1-litre volumetric flask, make up to volume with distilled water and mix.
Preparation and storage of standard solutions (procedure used for the validated method).
All standard solutions must be stored at < 5 °C and be prepared freshly on a monthly basis. Masses of components and solutions should be recorded to the nearest 0,1 mg.
Standard solution — AU.K.
Pipette the following reagents into a 100-ml volumetric flask, containing approximately 60-ml ethanol solution (5.13) to minimise component evaporation, make up to volume with ethanol solution (5.13) and mix thoroughly. Record the weight of the flask, each component added and the total final weight of contents.
| Component | Volume (ml) |
|---|---|
| Methanol (5.2) | 3,0 |
| Propan-1-ol (5.3) | 3,0 |
| 2-methylpropan-1-ol (5.4) | 3,0 |
| 2-methylbutan-1-ol (5.6) | 3,0 |
| 3-methylbutan-1-ol (5.7) | 3,0 |
| Ethyl acetate (5.8) | 3,0 |
| Butan-1-ol (5.9) | 3,0 |
| Butan-2-ol (5.10) | 3,0 |
| Acetaldehyde (5.11) | 3,0 |
| Acetal (5.12) | 3,0 |
Note 1:U.K.
It is preferable to add acetal and acetaldehyde last in order to minimise losses through evaporation.U.K.
Standard solution — BU.K.
Pipette 3 ml of pentan-3-ol, or other suitable internal standard, (5.5) into a 100-ml volumetric flask, containing approximately 80 ml ethanol solution (5.13), make up to volume with ethanol solution (5.13) and mix thoroughly.
Record the weight of the flask, the weight of pentan-3-ol or other internal standard added and the total final weight of contents.
Standard solution — CU.K.
Pipette 1 ml solution A (5.14.1) and 1 ml solution B (5.14.2) into a 100-ml volumetric flask containing approximately 80 ml ethanol solution (5.13), make up to volume with ethanol solution (5.13) and mix thoroughly.
Record the weight of the flask, each component added and the total final weight of contents.
Standard solution — DU.K.
In order to maintain analytical continuity, prepare a quality control standard using the previously prepared standard A (5.14.1). Pipette 1 ml solution A (5.14.1) into a 100-ml volumetric flask containing approximately 80 ml ethanol solution (5.13), make up to volume with ethanol solution (5.13) and mix thoroughly.
Record the weight of the flask, each component added and the total final weight of contents.
Standard solution — EU.K.
Pipette 10 ml solution B (5.14.2) into a 100-ml volumetric flask containing approximately 80 ml ethanol solution (5.13), make up to volume with ethanol solution (5.13) and mix thoroughly.
Record the weight of the flask, each component added and the total final weight of contents.
Standard solutions used to check the linearity of response of FIDU.K.
Into separate 100-ml volumetric flasks, containing approximately 80 ml ethanol (5.13), pipette 0, 0,1, 0,5, 1,0, 2,0 ml solution A (5.14.1) and 1 ml solution B (5.14.2), make up to volume with ethanol solution (5.13) and mix thoroughly.
Record the weight of the flask, each component added and the total final weight of contents.
6.Apparatus and equipmentU.K.
Apparatus capable of measuring the density and alcoholic strength.
Analytical balance, capable of measuring to four decimal places.
A temperature programmed gas chromatograph fitted with a flame ionisation detector and integrator or other data handling system capable of measuring peak areas or peak heights.
Gas chromatographic column(s), capable of separating the analytes such that the minimum resolution between the individual components (other than 2-methylbutan-1-ol and 3-methylbutan-1-ol) is at least 1.3.
Note 2:U.K.
The following columns and GC conditions are suitable examples:U.K.
A retention gap 1 m × 0,32 mm i.d. connected to a CP-WAX 57 CB column 50 m × 0,32 mm i.d. 0,2 μm film thickness (stabilised polyethylene glycol) followed by a Carbowax 400 column 50 m × 0,32 mm i.d. 0,2 μm film thickness. (Columns are connected using press-fit connectors.)
| Carrier gas and pressure: | Helium (135 kPa) |
| Column temperature: | 35 °C for 17 min., 35 to 70 °C at 12 °C/min., hold at 70 °C for 25 min. |
| Injector temperature: | 150 °C |
| Detector temperature: | 250 °C |
| Injection volume: | 1 μl, split 20 to 100:1 |
A retention gap 1 m × 0,32 mm i.d. connected to a CP-WAX 57 CB column 50 m × 0,32 mm i.d. 0,2 μm film thickness (stabilised polyethylene glycol). (Retention gap is connected using a press-fit connector.)
| Carrier gas and pressure: | Helium (65 kPa) |
| Column temperature: | 35 °C for 10 min., 35 to 110 °C at 5 °C/min., 110 to 190 °C at 30 °C/min., hold at 190 °C for 2 min. |
| Injector temperature: | 260 °C |
| Detector temperature: | 300 °C |
| Injection volume: | 1 μl, split 55:1 |
A packed column (5 % CW 20M, Carbopak B), 2 m × 2 mm i.d.
| Column temperature: | 65 °C for 4 min., 65 to 140 °C at 10 °C/min., hold at 140 °C for 5 min., 140 to 150 °C at 5 °C/min., hold at 150 °C for 3 min. |
| Injector temperature: | 65 °C |
| Detector temperature: | 200 °C |
| Injection volume: | 1 μl |
7.Sampling and samples.U.K.
Laboratory sample
On receipt, the alcoholic strength of each sample is measured (6.1).
8.Procedure (used for the validated method)U.K.
Test portion
Weigh an appropriate sealed weighing vessel and record the weight.
Pipette 9 ml laboratory sample into the vessel and record the weight (MSAMPLE).
Add 1 ml of standard solution E (5.14.5) and record the weight (MIS).
Shake the test material vigorously (at least 20 inversions). Samples must be stored at less than 5 °C prior to analysis in order to minimise any volatile losses.
Blank testU.K.
Using a four decimal place balance (6.2), weigh an appropriate sealed weighing vessel and record the weight.
Pipette 9 ml 400 ml/l ethanol solution (5.13) into the vessel and record the weight.
Add 1 ml of standard solution E (5.14.5) and record the weight.
Shake the test material vigorously (at least 20 inversions). Samples must be stored at less than 5 °C prior to analysis in order to minimise any volatile losses.
Preliminary testU.K.
Inject standard solution C (5.14.3) to ensure that all of the analytes are separated with a minimum resolution of 1.3 (except 2-methylbutan-1-ol and 3-methylbutan-1-ol).
CalibrationU.K.
The calibration should be checked using the following procedure. Ensure that the response is linear by successively analysing in triplicate each of the linearity standard solutions (5.14.6) containing internal standard (IS). From the integrator peak areas or peak heights for each injection calculate the ratio R for each congener and plot a graph of R versus the concentration ratio of congener to internal standard (IS), C. A linear plot should be obtained, with a correlation coefficient of at least 0,99. 
9.CalculationU.K.
An automated system of data handling can be used, provided the data can be checked using the principles described in the method below.
Measure either peak areas or peak heights for congener and internal standard peaks.
Response factor calculation.U.K.
From the chromatogram of the injection of standard solution C (5.14.3), calculate response factors for each congener using equation (1). 
=
Internal Standard
=
concentration of congener in solution C (5.14.3)
=
concentration of internal standard in solution C (5.14.3).
Sample analysisU.K.
Using equation (2) below, calculate the concentration of each congener in the samples. 
=
weight of sample (8.1.2);
=
weight of internal standard (8.1.3);
=
concentration of internal standard in solution E (5.14.5);
=
response factor calculated using equation 1.
10.Quality assurance and control (used for the validated method)U.K.
Using equation (2) above, calculate the concentration of each congener in the quality control standard solutions prepared by following the procedure as in 8.1.1 to 8.1.4. Using equation (3), calculate the percentage recovery of the target value. If the analysed results are within ± 10 % of their theoretical values for each congener, analysis may proceed. If not, an investigation should be made to find the cause of the inaccuracy and remedial action taken as appropriate.
11.Method performance characteristics (precision)U.K.
Statistical results of the interlaboratory test: the following tables give the values for the following compounds: ethanal, ethyl acetate, acetal, total ethanal, methanol, butan-2-ol, propan-1-ol, butan-1-ol, 2-methyl-propan-1-ol, 2 methyl-butan-1-ol, 3 methyl-butan-1-ol.
The following data were obtained from an international method performance study carried out to internationally agreed procedures.
| Year of interlaboratory test | 1 997 |
| Number of laboratories | 32 |
| Number of samples | 5 |
| Analyte | ethanal |
| Year of interlaboratory test | 1 997 |
| Number of laboratories | 32 |
| Number of samples | 5 |
| Analyte | ethyl acetate |
| Year of interlaboratory test | 1 997 |
| Number of laboratories | 32 |
| Number of samples | 5 |
| Analyte | acetal |
| Year of interlaboratory test | 1 997 |
| Number of laboratories | 32 |
| Number of samples | 5 |
| Analyte | total ethanal |
| Year of interlaboratory test | 1 997 |
| Number of laboratories | 32 |
| Number of samples | 5 |
| Analyte | Methanol |
| Year of interlaboratory test | 1 997 |
| Number of laboratories | 32 |
| Number of samples | 4 |
| Analyte | butan-2-ol |
| Year of interlaboratory test | 1 997 |
| Number of laboratories | 32 |
| Number of samples | 5 |
| Analyte | propan-1-ol |
| Year of interlaboratory test | 1 997 |
| Number of laboratories | 32 |
| Number of samples | 5 |
| Analyte | propan-1-ol |
| Year of interlaboratory test | 1 997 |
| Number of laboratories | 32 |
| Number of samples | 5 |
| Analyte | 2-methylpropan-1-ol |
| Year of interlaboratory test | 1 997 |
| Number of laboratories | 32 |
| Number of samples | 5 |
| Analyte | 2-methyl-butan-1-ol |
| Year of interlaboratory test | 1 997 |
| Number of laboratories | 32 |
| Number of samples | 5 |
| Analyte | 3-methyl-butan-1-ol |
[F3III.3. DETERMINATION OF VOLATILE ACIDITY OF SPIRIT DRINKS U.K.
1. Scope U.K.
The method has been validated in an interlaboratory study for rum, brandy, marc and fruit spirits, at levels ranging from 30 mg/l to 641 mg/l.
2. Normative references U.K.
ISO 3696: 1987 Water for analytical use — Specifications and test methods.
3. Definitions U.K.
3.1. Volatile acidity is calculated by deducting the fixed acidity from the total acidity. U.K.
3.2. Total acidity is the sum of titratable acidities. U.K.
3.3. Fixed acidity is the acidity of the residue left after evaporating the spirit to dryness. U.K.
4. Principle U.K.
The total acidity and fixed acidity are determined by titration or by potentiometry.
5. Reagents and materials U.K.
During the analysis, unless otherwise stated, use only reagents of recognised analytical grade and water of at least grade 3 as defined in ISO 3696:1987.
5.1. 0,01 M sodium hydroxide solution (NaOH) U.K.
5.2. Mixed indicator solution: U.K.
Weigh 0,1 g of indigo carmine and 0,1 g of phenol red.
Dissolve in 40 ml water and make up to 100 ml with ethanol.
6. Apparatus and equipment U.K.
Indirect laboratory apparatus, grade A glassware and the following:
Water pump
Rotary evaporator or ultrasonic bath
Equipment for potentiometric titration (optional).
7. Sampling and samples U.K.
Samples are stored at room temperature prior to analysis.
8. Procedure U.K.
8.1. Total acidity U.K.
8.1.1. Preparation of sample U.K.
The spirit is irradiated with ultrasonic (ultrasonication) or stirred two minutes under a vacuum to rid it of carbon dioxide if required.
8.1.2. Titration U.K.
Pipette 25 ml of the spirit into a 500 ml Erlenmeyer flask.
Add about 200 ml of cooled boiled distilled water (prepared fresh daily) and 2-6 drops of the mixed indicator solution (5.2).
Titrate with the 0,01 M sodium hydroxide solution (5.1) until the yellow-green colour changes to violet in the case of colourless spirits, the yellow-brown colour to red-brown in the case of brown-coloured spirits respectively.
The titration may also be carried out by potentiometry, to pH 7,5.
Let n 1 ml be the volume of the 0,01 M sodium hydroxide solution added.
8.1.3. Calculation U.K.
The total acidity (TA) expressed in milliequivalents per l of spirit is equal to 0,4 × n 1 .
The total acidity (TA′) expressed in mg of acetic acid per l of spirit is equal to 24 × n 1 .
8.2. Fixed acidity U.K.
8.2.1. Preparation of sample U.K.
Evaporate 25 ml of the spirit to dryness:
Pipette 25 ml of the spirit into a flat-bottomed cylindrical evaporating dish 55 mm in diameter. During the first hour of evaporation the evaporating dish is placed on the lid of a boiling water bath so that the liquid will not boil, as this could lead to losses through splattering.
Complete the drying by placing the evaporating dish in a drying oven at 105 °C for two hours. Allow the evaporating dish to cool in a desiccator.
8.2.2. Titration U.K.
Dissolve the residue left after evaporating with cooled boiled distilled water (prepared fresh daily) and make up to a volume to circa 100 ml and add 2-6 drops of the mixed indicator solution (5.2).
Titrate with the 0,01 M sodium hydroxide solution (5.1).
The titration may also be carried out by potentiometry, to pH 7,5.
Let n 2 ml be the volume of the 0,01 M sodium hydroxide solution added.
8.2.3. Calculation U.K.
The fixed acidity (FA) expressed in milliequivalents per l of spirit is equal to 0,4 × n 2 .
The fixed acidity (FA) expressed in mg of acetic acid per l of spirit is equal to 24 × n 2 .
9. Calculation of volatile acidity U.K.
9.1. Expression in milliequivalents per l: U.K.
Let:
=
total acidity in milliequivalents per l
=
fixed acidity in milliequivalents per l
Volatile acidity, VA, in milliequivalents per l is equal to:
TA – FA.
9.2. Expression in mg of acetic acid per l: U.K.
Let:
=
total acidity in mg of acetic acid per l
=
fixed acidity in mg of acetic acid per l
Volatile acidity, VA, in mg of acetic acid per l is equal to:
TA′ – FA′.
9.3. Expression in g of acetic acid per hl of pure 100 % vol. alcohol is equal to:  U.K.
U.K.
where A is the alcoholic strength by volume of the spirit drink.
10. Method performance characteristics (Precision) U.K.
10.1. Statistical results of the interlaboratory test U.K.
The following data were obtained from an international method performance study carried out to internationally agreed procedures (1) (2).
| Year of interlaboratory test | 2000 |
| Number of laboratories | 18 |
| Number of samples | 6 |
Sample types:
Plum spirit; split level *
Rum I; blind duplicates
Rum II; split level *
Slivovitz; blind duplicates
Brandy; blind duplicates
Marc spirit; blind duplicates.
(1) ‘Protocol for the design, conduct and interpretation of method-performance studies’, Horwitz, W. (1995) Pure and Applied Chemistry , 67, 332-343. U.K.
(2) Horwitz, W. (1982) Analytical Chemistry , 54, 67A-76A.] U.K.
Textual Amendments
[F4V. ANETHOLE. GAS CHROMATOGRAPHIC DETERMINATION OF TRANS-ANETHOLE IN SPIRIT DRINKS U.K.
1. Scope U.K.
This method is suitable for the determination of trans-anethole in aniseed-flavoured spirit drinks using capillary gas chromatography.
2. Normative references U.K.
ISO 3696: 1987 Water for analytical laboratory use — Specifications and test methods.
3. Principle U.K.
The trans-anethole concentration of the spirit is determined by gas chromatography (GC). The same quantity of an internal standard, e.g. 4-allylanisole (estragole) when estragole is not naturally present in the sample, is added to the test sample and to a trans-anethole reference solution of known concentration, both of which are then diluted with a 45 % ethanol solution and injected directly into the GC system. An extraction is necessary before sample preparation and analysis for liqueurs that contain large amounts of sugars.
4. Reagents and materials U.K.
During the analysis, use only reagents of a purity of at least 98 %. Water of at least grade 3 as defined by ISO 3696 should be used.
Reference chemicals should be stored cold (at 4 o C), away from light, in aluminium containers or in tinted (amber) glass reagent bottles. The stoppers should preferably be fitted with an aluminium seal. Trans-anethole will need to be ‘thawed’ from its crystalline state before use, but in this case its temperature should never exceed 35 o C.
4.1. Ethanol 96 % vol. (CAS 64-17-5) U.K.
4.2. 1-methoxy-4-(1-propenyl) benzene; (trans-anethole) (CAS 4180-23-8) U.K.
4.3. 4-allylanisole, (estragole) (CAS 140-67-0), suggested internal standard (IS) U.K.
4.4. Ethanol 45 % vol. U.K.
Add 560 g of distilled water to 378 g of ethanol 96 % vol.
4.5. Preparation of standard solutions U.K.
All standard solutions should be stored at room temperature (15 to 35 o C) away from light in aluminium containers or in tinted (amber) glass reagent bottles. The stopper should preferably be fitted with an aluminium seal.
Trans-anethole and 4-allylanisole are practically insoluble in water, and it is therefore necessary to dissolve the trans-anethole and 4-allylanisole in some 96 % ethanol (4.1) before the addition of 45 % ethanol (4.4).
The stock solutions must be freshly prepared each week.
4.5.1. Standard solution A U.K.
Stock solution of trans-anethole (concentration: 2 g/l)
Weigh 40 mg of trans-anethole (4.2) in a 20 ml volumetric flask (or 400 mg in 200 ml, etc.). Add some 96 % ethanol (4.1) and make up to volume with 45 % vol. ethanol (4.4), mix thoroughly.
4.5.2. Internal standard solution B U.K.
Stock solution of internal standard, e.g. estragole (concentration: 2 g/l)
Weigh 40 mg of estragole (4.3) in a 20 ml volumetric flask (400 mg in 200 ml etc.). Add some 96 % vol. ethanol (4.1) make up to volume with 45 % vol. ethanol (4.4), mix thoroughly.
4.5.3. Solutions used to check the linearity response of the flame ionisation detector (FID) U.K.
The linearity response of the FID must be checked for the analysis taking into account a range of concentrations of trans-anethole in spirits from 0 g/l up to 2.5 g/l. In the procedure of analysis, the unknown samples of spirits to be analysed are diluted 10 times (8.3). For the conditions of the analysis described in the method, stock solutions corresponding to concentrations of 0, 0,05, 0,1, 0,15, 0,2, and 0,25 g/l of trans-anethole in the sample to be analysed are prepared as follows: take 0,5, 1, 1,5, 2, and 2,5 ml of stock solution A (4.5.1) and pipette in separate 20 ml volumetric flasks; pipette into each flask 2 ml of internal standard solution B (4.5.2) and make up to volume with 45 % vol. ethanol (4.4), mix thoroughly.
The blank solutions (8.4) is used as the 0 g/l solution.
4.5.4. Standard solution C U.K.
Take 2 ml of standard solution A (4.5.1) and pipette into a 20 ml volumetric flask, then add 2 ml of internal standard solution B (4.5.2) and make up to volume with 45 % vol. ethanol (4.4), mix thoroughly.
5. Apparatus and equipment U.K.
5.1. A capillary gas chromatograph fitted with a flame ionisation detector (FID) and integrator or other data handling system capable of measuring peak heights or areas, and with an automatic sampler or the necessary equipment for manual sample injection. U.K.
5.2. Split/splitless injector U.K.
5.3. Capillary column, for example: U.K.
Length: 50 m
Internal diameter: 0,32 mm
Film thickness: 0,2 μm
Stationary phase: FFAP — modified TPA polyethylene glycol cross-linked porous polymer.
5.4. Common laboratory equipment: A grade volumetric glassware, analytical balance (precision: ± 0,1 mg). U.K.
6. Chromatography conditions U.K.
The column type and dimensions, and the GC conditions, should be such that anethole and the internal standard are separated from each other and from any interfering substances. Typical conditions for the column given as an example in 5.3 are:
Carrier gas: analytical helium
Flow rate: 2 ml/min
Injector temperature: 250 o C
Detector temperature: 250 o C
Oven temperature conditions: isothermal, 180 o C, run time 10 minutes
Injection volume: 1 μl, split 1:40.
7. Samples U.K.
Samples should be stored at room temperature, away from light and cold.
8. Procedure U.K.
8.1. Sample screening for estragole U.K.
To ensure that there is no estragole naturally present in the sample, a blank analysis should be carried out without the addition of any internal standard. If estragole is naturally present then another internal standard must be chosen (for instance menthol).
Pipette 2 ml sample into a 20 ml volumetric flask and make up to volume with 45 % vol. ethanol (4.4), mix thoroughly.
8.2. Preparation of unknown samples U.K.
Pipette 2 ml sample into a 20 ml volumetric flask then add 2 ml of internal standard solution B (4.5.2) and make up to volume with 45 % vol. ethanol (4.4), mix thoroughly.
8.3. Blank U.K.
Pipette 2 ml of internal standard solution B (4.5.2) into a 20 ml volumetric flask and make up to volume with 45 % vol. ethanol (4.4), mix thoroughly.
8.4. Linearity test U.K.
Prior to the commencement of the analysis the linearity of the response of the FID should be checked by successively analysing in triplicate each of the linearity standard solutions (4.5.3).
From the integrator peak areas or peak heights for each injection plot a graph of their mother solution concentration in g/l versus the ratio R for each.
=
trans-anethole peak height or area divided by the estragole peak height or area.
A linear plot should be obtained.
8.5. Determination U.K.
Inject the blank solution (8.3), followed by standard solution C (4.5.4), followed by one of the linearity standards (4.5.3) which will act as a quality control sample (this may be chosen with reference to the probable concentration of trans-anethole in the unknown), followed by five unknowns (8.2); insert a linearity (quality control) sample after every five unknown samples, to ensure analytical stability.
9. Calculation of response factor U.K.
Measure either peak areas (using an integrator or other data system) or peak heights (manual integration) for trans-anethole and internal standard peaks.
9.1. Response factor (RF i ) calculation U.K.
The response factor is calculated as follows
where:
is the concentration of trans-anethole in the standard solution A (4.5.1)
is the concentration of internal standard in the standard solution B (4.5.2)
is the area (or height) of the trans-anethole peak
the area (or height) of the internal standard peak
RF i is calculated from the five samples of solution C (4.5.4).
9.2. Analysis of the linearity response test solutions U.K.
Inject the linearity response test solutions (4.5.3).
9.3. Analysis of the sample U.K.
Inject the unknown sample solution (8.2).
10. Calculation of results U.K.
The formula for the calculation of the concentration of trans-anethole is the following:
where:
is the unknown trans-anethole concentration
is the concentration of internal standard in the unknown (4.5.2)
is the area or height of the trans-anethole peak
the area or height of the internal standard peak
is the response coefficient (calculated as in 9.1)
The trans-anethole concentration is expressed as grams per litre, to one decimal place.
11. Quality assurance and control U.K.
The chromatograms should be such that anethole and the internal standard are separated from each other and from any interfering substances. The RF i value is calculated from the results for the five injections of solution C (4.5.4). If the coefficient of variation (CV % = (standard deviation/mean)*100)) is within plus or minus 1 %, the RF i average value is acceptable.
The calculation above should be used to calculate the concentration of trans-anethole in the sample selected for the quality control from the linearity control solutions (4.5.3).
If the mean calculated results from analysis of the linearity solution selected for internal quality control sample (IQC) are within plus or minus 2,5 % of their theoretical value, then the results for the unknown samples can be accepted.
12. Treatment of spirits sample containing large amount of sugar and of liqueur sample prior to GC analysis U.K.
Extraction of alcohol from spirit drink containing a large amount of sugar, in order to be able to determine the trans-anethole concentration using capillary gas chromatography.
12.1. Principle U.K.
An aliquot of the liqueur sample is taken and to this is added the internal standard, at a concentration similar to that of the analyte (trans-anethole) in the liqueur. To this are added sodium phosphate dodecahydrate and anhydrous ammonium sulphate. The resulting mixture is well shaken and chilled, two layers develop, and the upper alcohol layer is removed. An aliquot of this alcohol layer is taken and diluted with 45 % ethanol solution (4.4) (Note: no internal standard is added at this stage, because it has already been added). The resulting solution is analysed in gas chromatography.
12.2. Reagents and materials U.K.
During the extraction use only reagents of a purity greater than 99 %.
12.2.1. Ammonium sulphate, anhydrous, (CAS 7783-20-2). U.K.
12.2.2. Sodium phosphate, dibasic, dodecahydrate, (CAS 10039-32-4). U.K.
12.3. Apparatus and equipment U.K.
Conical flasks, separating flasks, refrigerator.
12.4. Procedure U.K.
12.4.1. Sample screening for estragole U.K.
To ensure that there is no estragole naturally present in the sample, a blank extraction (12.6.2) and analysis should be carried out without the addition of any internal standard. If estragole is naturally present then another internal standard must be chosen.
12.4.2. Extraction U.K.
Pipette 5 ml of 96 % ethanol (4.1) into a conical flask, weigh into this flask 50 mg of internal standard (4.3), and add 50 ml of the sample. Add 12 g of ammonium sulphate, anhydrous (12.2.1), and 8.6 g of dibasic sodium phosphate, dodecahydrate (12.2.2). Stopper the conical flask.
Shake the flask for at least 30 minutes. A mechanical shaking device may be used, but not a Teflon coated magnetic stirring bar, as the Teflon will absorb some of the analyte. Note that the added salts will not dissolve completely.
Place the stoppered flask in a refrigerator (T < 5 C) for at least two hours.
After this time, there should be two distinct liquid layers and a solid residue. The alcohol layer should be clear; if not, replace in the refrigerator until a clear separation is achieved.
When the alcohol layer is clear, carefully take an aliquot (e.g. 10 ml), without disturbing the aqueous layer, place in an amber vial and close securely.
12.4.3. Preparation of the extracted sample to be analysed U.K.
Allow extract (12.4.2) to reach room temperature.
Take 2 ml of the alcohol layer of the attemperated extracted sample and pipette into a 20 ml volumetric flask, make up to volume with 45 % ethanol (4.4), mix thoroughly.
12.5. Determination U.K.
Follow the procedure as outlined in 8.5.
12.6. Calculation of results U.K.
Use the following formula to calculate the results:
where:
is the weight of internal standard (4.3) taken (12.4.2) (in milligrams)
is the volume of unknown sample (50 ml)
is the response factor (9.1)
is the area of the trans-anethole peak
is the area of the internal standard peak
The results are expressed in grams per litre, to one decimal place.
12.7. Quality control and assurance U.K.
Follow the procedure as outlined in 11 above.
13. Method performance characteristics (precision) U.K.
Statistical results of the interlaboratory test:
the following tables give the values for anethole.
The following data were obtained from an international method performance study carried out to internationally agreed procedures.
Year of interlaboratory test 1998 Number of laboratories 16 Number of samples 10 Analyte anethole Pastis:
Samples A B C D E F Number of laboratories retained after eliminating outliers 15 15 15 13 16 16 Number of outliers (laboratories) 1 1 1 3 — — Number of accepted results 30 30 30 26 16 16 Mean value g/l 1,477 1,955 1,94 1,833 1,741 1,754 Repeatability standard deviation (S r ) g/l 0,022 0,033 0,034 0,017 — — Repeatability relative standard deviation (RSD r ) (%) 1,5 1,7 1,8 0,9 — — Repeatability limit (r) g/l 0,062 0,093 0,096 0,047 — — Reproducibility standard deviation (S R ) g/l 0,034 0,045 0,063 0,037 0,058 0,042 Reproducibility relative standard deviation (RSD R ) (%) 2,3 2,3 3,2 2,0 3,3 2,4 Reproducibility limit (R) g/l 0,094 0,125 0,176 0,103 0,163 0,119 Sample types:
Apastis, blind duplicates
Bpastis, blind duplicates
Cpastis, blind duplicates
Dpastis, blind duplicates
Epastis, single duplicates
Fpastis, single duplicates
Other aniseed-flavoured spirit drinks:
Samples G H I J Number of laboratories retained after eliminating outliers 16 14 14 14 Number of outliers (Laboratories) — 2 1 1 Number of accepted results 32 28 28 28 Mean value g/l 0,778
0,530 (*)
1,742 0,351 0,599 Repeatability standard deviation (S r ) g/l 0,02 0,012 0,013 0,014 Repeatability relative standard deviation (RSD r ) (%) 3,1 0,7 3,8 2,3 Repeatability limit (r) g/l 0,056 0,033 0,038 0,038 Reproducibility standard deviation (S R ) g/l 0,031 0,029 0,021 0,03 Repeatability relative standard deviation (RSD R ) (%) 4,8 1,6 5,9 5,0 Reproducibility limit (R) g/l 0,088 0,08 0,058 0,084 Sample types:
Gouzo, split levels (*)
Hanis, blind duplicates
Ianiseed-flavoured liqueur, duplicates
Janiseed-flavoured liqueur, duplicates.
Textual Amendments
VI. GLYCYRRHIZIC ACID. DETERMINATION OF GLYCYRRHIZIC ACID USING HIGH PERFORMANCE LIQUID CHROMATOGRAPHY U.K.
1. Scope U.K.
This method is suitable for the determination of glycyrrhizic acid in aniseed-flavoured spirit drinks using high performance liquid chromatography (HPLC). [F5Regulation 110/2008] specifies that any aniseed-flavoured spirit called ‘ pastis ’ must contain between 0,05 and 0,5 g of glycyrrhizic acid per litre.
Textual Amendments
F5Words in Annex Ch. 6 para. 1 substituted (31.12.2020) by The Agricultural Products, Food and Drink (Amendment etc.) (EU Exit) Regulations 2020 (S.I. 2020/1637), regs. 1(7), 16(6)(b)
2. Normative references U.K.
ISO 3696: 1987 Water for analytical laboratory use — Specifications and test methods.
3. Principle U.K.
The glycyrrhizic acid concentration is determined using high-performance liquid chromatography (HPLC) with UV detection. A standard solution and the test sample are filtered and they are separately injected directly into the HPLC system.
4. Reagents and materials U.K.
During the analysis, use only reagents of HPLC grade, absolute ethanol and water of grade 3 as defined by ISO 3696.
4.1. Ethanol 96 % vol. (CAS 64-17-5). U.K.
4.2. Ammonium glycyrrhizinate, C 42 H 62 O 16 .NH 3 (Glycyrrhizic acid ammonium salt) U.K.
(Mol. Wt.: 839,98)(CAS 53956-04-0): purity at least 90 %
(Mol. Wt.: glycyrrhizic acid 822,94).
4.3. Glacial acetic acid, CH 3 COOH, (CAS 64-19-7). U.K.
4.4. Methanol, CH 3 OH (CAS 67-56-1). U.K.
4.5. Ethanol 50 % vol. U.K.
For 1 000 ml at 20 o C:
96 % vol. ethanol (4.1): 521 ml
Water (2.0): 511 ml.
4.6. Preparation of the HPLC elution solutions U.K.
4.6.1. Elution solvent A (example) U.K.
80 parts (by volume) of water (2.0)
20 parts (by volume) of acetic acid (4.3).
Degas the elution solvent for five minutes.
Note: U.K.
If the water being used has not been microfiltered, it is advisable to filter the prepared elution solvent on a filter for organic solvents with a pore size less than or equal to 0,45 μm. U.K.
4.6.2. Elution solvent B U.K.
Methanol (4.4).
4.7. Preparation of standard solutions U.K.
All standard solutions must be freshly prepared after two months.
4.7.1. Reference solution C U.K.
Weigh to the nearest 0,1 mg, 25 mg of ammonium glycyrrhizinate (4.2) in a 100 ml volumetric flask. Add some 50 % vol. ethanol (4.5) and dissolve the ammonium glycyrrhizinate. When it has dissolved make up to the mark with 50 % vol. ethanol (4.5).
Filter through a filter for organic solvents.
4.7.2. Standard solutions used to check the linearity of the response of the instrumentation U.K.
A 1,0 g/l stock solution is prepared by weighing, to the nearest 0,1 mg, 100 mg of ammonium glycyrrhizinate in a 100 ml volumetric flask. Add some 50 % vol. ethanol (4.5) and dissolve the ammonium glycyrrhizinate. When it has dissolved make up to the mark with 50 % vol. ethanol (4.5).
At least four other solutions corresponding to 0,05, 0,1, 0,25 and 0,5 g/l of ammonium glycyrrhizinate are prepared by pipetting respectively 5 ml, 10 ml, 25 ml and 50 ml of the 1,0 g/l stock solution in separate 100 ml volumetric flasks. Then make up to the mark with 50 % vol. ethanol (4.5) and mix up thoroughly.
Filter all solutions though a filter for organic solvents.
5. Apparatus and equipment U.K.
5.1. Separation system U.K.
5.1.1. High-performance liquid chromatograph. U.K.
5.1.2. Pumping system enabling one to achieve and maintain a constant or programmed rate of flow with great precision. U.K.
5.1.3. UV spectrophotometric detection system: to be set at 254 nm. U.K.
5.1.4. Solvent degassing system. U.K.
5.2. Computational integrator or recorder, the performance of which is compatible with the rest of the system. U.K.
5.3. Column (example): U.K.
Material: stainless steel or glass
Internal diameter: 4 to 5 mm
Length: 100 to 250 mm
Stationary phase: cross-linked silica with a (preferably spherical) octadecyl functional group (C18), maximum particle size: 5 μm.
5.4. Laboratory equipment U.K.
5.4.1. Analytical balance with a precision of 0,1 mg U.K.
5.4.2. A-grade volumetric glassware U.K.
5.4.3. Micromembrane filtration arrangement for small volumes. U.K.
6. Chromatography conditions U.K.
6.1. Elution characteristics: (example) U.K.
flow rate: 1 ml/minute,
solvent A = 30 %,
solvent B = 70 %.
6.2. Detection: U.K.
UV = 254 nm
7. Procedure U.K.
7.1. Preparation of the spirit sample U.K.
Filter, if necessary, through a filter for organic solvents (pore diameter: 0,45 μm).
7.2. Determination U.K.
Once the chromatography conditions have stabilised,
inject 20 μl of the reference solution C (4.7.1),
inject 20 μl of the sample solution,
compare the two chromatograms. Identify the glycyrrhizic acid peaks from their retention times. Measure their areas (or heights) and calculate the concentration in g/l to two decimal figures using the following equation:
where:
cis the concentration in grams per litre of glycyrrhizic acid in the spirit being analysed
Cis the concentration in grams per litre of ammonium glycyrrhizinate in the reference solution
his the area (or height) of the glycyrrhizic acid peak of the spirit being analysed
His the area (or height) of the glycyrrhizic acid peak of the reference solution
Pis the purity of the reference ammonium glycyrrhizinate (in %)
823is the mass of one mole of glycyrrhizic acid
840is the mass of one mole of ammonium glycyrrhizinate.
8. Method performance characteristics (precision) U.K.
Statistical results of the interlaboratory test:
the following table give the values for glycyrrhizic acid.
The following data were obtained from an international method performance study carried out to internationally agreed procedures.
Year of interlaboratory test 1998 Number of laboratories 16 Number of samples 5 Analyte glycyrrhizic acid Samples A B C D F Number of laboratories retained after eliminating outliers 13 14 15 16 16 Number of outliers (laboratories) 3 2 1 — — Number of accepted results 26 28 30 32 32 Mean value g/l 0,046 0,092
(*) 0,099
0,089 0,249 0,493 Repeatability standard deviation (S r ) g/l 0,001 0,001 0,001 0,002 0,003 Repeatability relative standard deviation (RSD r ) (%) 1,5 1,3 0,7 1,0 0,6 Repeatability limit (r) g/l 0,002 0,004 0,002 0,007 0,009 Reproducibility standard deviation (S R ) g/l 0,004 0,007 0,004 0,006 0,013 Reproducibility relative standard deviation (RSD R ) (%) 8,6 7,2 4,0 2,5 2,7 Reproducibility limit (R) g/l 0,011 0,019 0,01 0,018 0,037 Sample types:
Apastis, blind duplicates
Bpastis, split levels (*)
Cpastis, blind duplicates
Dpastis, blind duplicates
Epastis, blind duplicates
VII. CHALCONES. HIGH-PERFORMANCE LIQUID CHROMATOGRAPHY METHOD FOR VERIFYING THE PRESENCE OF CHALCONES IN PASTIS U.K.
1. Scope U.K.
This method is suitable for determining whether chalcones are present in aniseed-flavoured drinks or not. Chalcones are natural colorants of the flavonoid family that are present in liquorice root ( Glycyrrhiza glabra ).
For an aniseed-flavoured spirit to be called ‘ pastis ’ , it must contain chalcones ([F6Regulation 110/2008]).
Textual Amendments
F6Words in Annex Ch. 7 para. 1 substituted (31.12.2020) by The Agricultural Products, Food and Drink (Amendment etc.) (EU Exit) Regulations 2020 (S.I. 2020/1637), regs. 1(7), 16(6)(c)
2. Normative references U.K.
ISO 3696: 1987, Water for analytical laboratory use — Specifications and test methods.
3. Principle U.K.
A reference liquorice extract solution is prepared. The presence or absence of chalcones is determined using high-performance liquid chromatography (HPLC) with UV detection.
4. Reagents and materials U.K.
During the analysis, use only reagents of HPLC grade. The ethanol should be 96 % vol. Only water of grade 3 as defined by ISO 3696 should be used.
4.1. Ethanol 96 % vol. (CAS 64-17-5) U.K.
4.2. Acetonitrile, CH 3 CN, (CAS 75-05-8) U.K.
4.3. Reference substance: Glycyrrhiza glabra : liquorice, ‘sweet root’U.K.
Coarsely ground liquorice roots ( Glycyrrhiza glabra ). Average dimensions of the rodlike particles: length: 10 to 15 mm, thickness: 1 to 3 mm.
4.4. Sodium acetate, CH 3 COONa, (CAS 127-09-3) U.K.
4.5. Glacial acetic acid, CH 3 COOH, (CAS 64-19-7) U.K.
4.6. Preparation of solutions U.K.
4.6.1. Ethanol 50 % volume U.K.
For 1 000 ml at 20 °C:
96 % vol. ethanol (4.1): 521 ml,
Water (2.0): 511 ml.
4.6.2. Solvent A: acetonitrile U.K.
Acetonitrile (4.2) of HPLC analytical purity.
Degas
4.6.3. Solvent B: 0,1 M sodium acetate buffer solution, pH 4,66. U.K.
Weigh 8,203 g of sodium acetate (4.4), add 6,005 g of glacial acetic acid (4.5) and make up to 1 000 ml with water (2) in a volumetric flask.
5. Preparation of the reference extract from Glycyrrhiza glabra (4.3) U.K.
5.1. Weigh 10 g of ground liquorice root ( Glycyrrhiza glabra ) (4.3) and place in a round-bottomed distillation flask U.K.
add 100 ml of 50 % vol. ethanol (4.6.1),
boil under reflux for one hour,
filter,
set the filtrate aside for later use.
5.2. Recover the liquorice extract from the filter U.K.
place in a round-bottomed distillation flask,
add 100 ml of 50 % vol. ethanol (4.6.1),
boil under reflux for one hour,
filter. Set aside the filtrate for later use.
5.3. The liquorice root extraction must be performed three times in succession. U.K.
5.4. Combine the three filtrates. U.K.
5.5. Evaporate the solvent phase (of 5.4) on a rotary evaporator. U.K.
5.6. Take up the residual extract (of 5.5) with 100 ml 50 % vol. ethanol (4.6.1). U.K.
6. Apparatus and equipment U.K.
6.1. Separation system. U.K.
6.1.1. High-performance liquid chromatograph. U.K.
6.1.2. Pumping system capable of achieving and maintaining a constant or programmed rate of flow at high pressure. U.K.
6.1.3. UV/visible spectrophotometric detection system that can be set at 254 and 370 nm. U.K.
6.1.4. Solvent degassing system: U.K.
6.1.5. Column oven that can be set at a temperature of 40 ± 0,1 °C. U.K.
6.2. Computational integrator or recorder, the performance of which is compatible with the rest of the separation system. U.K.
6.3. Column U.K.
Material: stainless steel or glass
Internal diameter: 4 to 5 mm
Stationary phase: cross-linked silica with an octadecyl derived functional group (C18), particle size: 5 μm at most (cross-linked phase).
6.4. Common laboratory equipment, including: U.K.
analytical balance. (precision: ± 0,1 mg);
distillation apparatus with a reflux condenser, comprising, for example:
a 250 ml round-bottomed flask with a standardised ground-glass joint,
a 30 cm long reflux condenser, and
a heat source (any pyrogenic reaction involving the extractive matter must be avoided by using an appropriate arrangement).
Rotary evaporation apparatus.
Filtration set-up (i.e. Buchner funnel).
6.5. Chromatography conditions (example). U.K.
6.5.1. Elution characteristics of solvents A (4.6.2) and B (4.6.3): U.K.
shift from 20/80 (v/v) to 50/50 (v/v) gradient in 15 minutes,
shift from 50/50 (v/v) to 75/25 (v/v) gradient in five minutes,
equal strength at 75/25 (v/v) for five minutes,
stabilisation of the column between injections,
equal strength at 20/80 (v/v) for five minutes.
6.5.2. Flow rate: 1 ml/minute. U.K.
6.5.3. UV detector settings: U.K.
the detector must be set at 370 nm to detect the presence of chalcones and then at 254 nm to detect glycyrrhizic acid.
Note: U.K.
the change of wavelength (from 370 nm to 254 nm) must be carried out 30 seconds before the beginning of the peak of elution of glycyrrhizic acid. U.K.
7. Procedure U.K.
7.1. Preparation of the spirit sample U.K.
Filter through a filter for organic solvents (pore diameter: 0,45 μm).
7.2. Preparation of the residual liquorice extract (5.6) U.K.
Make a one in ten dilution with 50 % vol. ethanol (4.6.1) before analysis.
7.3. Determination U.K.
7.3.1. Inject 20 μl of the prepared liquorice extract (7.2). Perform the analysis using the chromatography conditions described above (6.5). U.K.
7.3.2. Inject 20 μl of the sample (7.1) (aniseed-flavoured spirit sample). Perform the analysis using the chromatography conditions described above (6.5). U.K.
7.3.3. Compare the two chromatograms. There must be a great similarity between the two chromatograms in the chalcone exit zone (during the detection at 370 nm under the analysis conditions described above) (see Figure 1). U.K.
8. Characteristic chromatogram for a pastis U.K.
9. Method performance characteristics (precision) U.K.
Results of the interlaboratory test:
the following table gives the performance for recognition of presence or absence of chalcones in pastis and aniseed-flavoured spirits.
The following data were obtained from an international method performance study carried out to internationally agreed procedures.
Year of interlaboratory test 1998 Number of laboratories 14 Number of samples 11 Analyte chalcones aInconsistent results between the two duplicates, attributed to a sampling error
Samples A B C D E F Number of laboratories retained after eliminating outliers 14 14 14 14 14 13 Number of outliers (laboratories) — — — — — 1 a Number of accepted results 28 14 14 28 28 26 Number of results for presence of chalcones 28 14 14 0 28 0 Number of results for absence of chalcones 0 0 0 28 0 26 Percentage of correct results (%) 100 100 100 100 100 100 Samples G H I J K Number of laboratories retained after eliminating outliers 14 14 14 14 14 Number of outliers (laboratories) — — — — — Number of accepted results 28 14 14 28 28 Number of results for presence of chalcones 0 0 0 0 0 Number of results for absence of chalcone 28 14 14 28 28 Percentage of correct results (%) 100 100 100 100 100 Sample types:
Apastis, blind duplicates
Bpastis, single sample
Cpastis, single sample
D‘ pastis ’ (not containing chalcones), blind duplicates
E‘ pastis ’ (not containing chalcones), blind duplicates
Faniseed-flavoured liqueur (not containing chalcones), blind duplicates
Ganiseed-flavoured liqueur (not containing chalcones), blind duplicates
Houzo (not containing chalcones), single sample
Iouzo (not containing chalcones), single sample
Janis (not containing chalcones), blind duplicates
K‘ pastis ’ (not containing chalcones), blind duplicates.
[F3VIII. TOTAL SUGARS U.K.
1. Scope U.K.
The HPLC–RI method is applicable for the determination of total sugars (expressed as invert sugar) in spirit drinks, with the exclusion of liqueurs containing egg and milk products.
The method has been validated in an interlaboratory study for pastis, distilled anis, cherry liqueur, crème de (followed by the name of a fruit or the raw material used) and crème de cassis, at levels ranging from 10,86 g/l to 509,7 g/l. However, linearity of the instrument response was proven for the concentration range 2,5 g/l to 20,0 g/l.
This method is not intended for determining low levels of sugars.
2. Normative references U.K.
ISO 3696:1987 Waters for analytical use — Specifications and test methods.
3. Principle U.K.
High-performance liquid chromatography assays of sugar solutions, in order to determine their glucose, fructose, sucrose, maltose and lactose concentrations.
This method uses an alkylamine stationary phase and differential refractometry detection and is given as an example. The use of anion exchange resins as stationary phase would also be possible.
4. Reagents and materials U.K.
4.1. Glucose (CAS 50-99-7), at least 99 % pure. U.K.
4.2. Fructose (CAS 57-48-7), at least 99 % pure. U.K.
4.3. Sucrose (CAS 57-50-1), at least 99 % pure. U.K.
4.4. Lactose (CAS 5965-66-2), at least 99 % pure. U.K.
4.5. Maltose monohydrate (CAS 6363-53-7), at least 99 % pure. U.K.
4.6. Pure acetonitrile (CAS 75-05-8) for HPLC analysis. U.K.
4.7. Distilled or demineralised water, preferably microfiltered. U.K.
4.8. Solvents (example) U.K.
The elution solvent is composed of:
75 parts by volume of acetonitrile (4.6),
25 parts by volume of distilled water (4.7).
Pass helium through at a slow rate for 5-10 minutes prior to use to degas.
If the water being used has not been microfiltered, the solvent should be filtered with a filter for organic solvents with a pore size less than or equal to 0,45 μm.
4.9. Ethanol absolute (CAS 64-17-5). U.K.
4.10. Ethanol solution (5 %, v/v). U.K.
4.11. Preparation of stock standard solution (20 g/l) U.K.
Weigh 2 g each of the sugars to be analysed (4.1 to 4.5), transfer them without loss to a 100 ml volumetric flask. (NB 2,11 g of maltose monohydrate is equivalent to 2 g of maltose).
Adjust to 100 ml with a 5 % vol. alcohol solution (4.10), shake and store at around + 4 °C. Prepare a new stock solution once a week.
4.12. Preparation of working standard solutions (2,5, 5,0, 7,5, 10,0 and 20,0 g/L) U.K.
Dilute the stock solution, 20 g/l (4.11) appropriately with a 5 % vol. alcohol solution (4.10) to give five working standards of 2,5, 5,0, 7,5, 10,0 and 20,0 g/l. Filter with a filter of a pore size less than or equal to 0,45 μm (5.3).
5. Apparatus and Equipment U.K.
5.1. HPLC system capable of achieving baseline resolution of all of the sugars. U.K.
5.1.1. High-performance liquid chromatograph with a six-way injection valve fitted with a 10 μl loop or any other device, whether automatic or manual, for the reliable injection of microvolumes. U.K.
5.1.2. Pumping system enabling one to achieve and maintain a constant or programmed rate of flow with great precision. U.K.
5.1.3. Differential refractometer. U.K.
5.1.4. Computational integrator or recorder, the performance of which is compatible with the rest of the set-up. U.K.
5.1.5. Pre-column: U.K.
It is recommended that a suitable pre-column is attached to the analytical column.
5.1.6. Column (example): U.K.
| Material: | stainless steel or glass. |
| Internal diameter: | 2-5 mm. |
| Length: | 100-250 mm (depending on the packing particle size), for example, 250 mm if the particles are 5 μm in diameter. |
| Stationary phase: | alkylamine functional groups bonded to silica, maximum particle size 5 μm. |
5.1.7. Chromatography conditions (example): U.K.
Elution solvent (4.8), flow rate: 1 ml/minute.
Detection: Differential refractometry.
To make certain that the detector is perfectly stable, it should be switched on a few hours before use. The reference cell must be filled with the elution solvent.
5.2. Analytical balance accurate to 0,1 mg. U.K.
5.3. Filtration set-up for small volumes using a 0,45 μm micromembrane. U.K.
6. Sample storage U.K.
On receipt, samples are to be stored at room temperature prior to analysis.
7. Procedure U.K.
7.1. PART A: Sample preparation U.K.
7.1.1. Shake the sample. U.K.
7.1.2. Filter the sample through a filter with a pore size less than or equal to 0,45 μm (5.3). U.K.
7.2. PART B: HPLC U.K.
7.2.1. Determination U.K.
Inject 10 μl of the standard solutions (4.12) and samples (7.1.2). Perform the analysis under suitable chromatography conditions, for example those described above.
7.2.2. Should any peak of a sample have a greater area (or height) than the corresponding peak in the most concentrated standard, then the sample should be diluted with distilled water and reanalysed. U.K.
8. Calculation U.K.
Compare the two chromatograms obtained for the standard solution and spirit. Identify the peaks by their retention times. Measure their areas (or heights) to calculate the concentrations by the external standard method. Take into account any dilutions made to the sample.
The final result is the sum of sucrose, maltose, lactose, glucose and fructose, expressed as invert sugar in g/l.
Invert sugar is calculated as the sum of all monosaccharides and reducing disaccharides present, plus the stoichiometric amount of glucose and fructose calculated from the sucrose present.
=
glucose (g/l) + fructose (g/l) + maltose (g/l) + lactose (g/l) + (sucrose (g/l) × 1,05).
=
(molecular weight of fructose + molecular weight of glucose)/molecular weight of sucrose.
9. Method performance characteristics (precision) U.K.
9.1. Statistical results of the interlaboratory test U.K.
The following data were obtained from an international method performance study carried out to internationally agreed procedures ( 1 ) ( 2 ).
| Year of interlaboratory test | 2000 |
| Number of laboratories | 24 |
| Number of samples | 8 |
(1) ‘ Protocol for the design, conduct and interpretation of method-performance studies ’ , Horwitz, W. (1995) Pure and Applied Chemistry , 67, 332-343. U.K.
(2) Horwitz, W. (1982) Analytical Chemistry , 54, 67A-76A. U.K.
Table 1
Fructose, glucose, maltose
| Analyte | Fructose | Glucose | Maltose | ||||
|---|---|---|---|---|---|---|---|
| Samples (× 2) | Crème de Cassis | Standard (50 g/l) | Aniseed-flavoured spirit drink | Crème de Cassis | Standard (50 g/l) | Aniseed-flavoured spirit drink | Standard (10 g/l) |
| Mean value (g/l) | 92,78 | 50,61 | 15,62 | 93,16 | 50,06 | 15,81 | 9,32 |
| No of labs without outliers | 21 | 22 | 21 | 23 | 19 | 21 | 22 |
| Repeatability standard deviation, s r , (g/l) | 2,34 | 2,12 | 0,43 | 3,47 | 1,01 | 0,48 | 0,54 |
| Repeatability relative standard deviation, RSD r (%) | 2,53 | 4,2 | 2,76 | 3,72 | 2,03 | 3,02 | 5,77 |
| Repeatability limit, r (g/l) (r = 2,8 × s r ) | 6,56 | 5,95 | 1,21 | 9,71 | 2,84 | 1,34 | 1,51 |
| Reproducibility standard deviation, s R (g/l) | 7,72 | 3,13 | 0,84 | 9,99 | 2,7 | 0,88 | 1,4 |
| Reproducibility relative standard deviation, RSD R (%) | 8,32 | 6,18 | 5,37 | 10,72 | 5,4 | 5,54 | 15,06 |
| Reproducibility limit, R (g/l) (R = 2,8 × sR) | 21,62 | 8,76 | 2,35 | 27,97 | 7,57 | 2,45 | 3,93 |
Table 2
Sucrose
| a split level. | ||||||
| Analyte | Sucrose | |||||
|---|---|---|---|---|---|---|
| Samples | Pastis | Ouzo | Cherry liqueur | Crème de Menthe | Crème de Cassis | Standard (100 g/l) |
| Mean value (g/l) | 10,83 | 29,2 19,7 a | 103,33 | 349,96 | 319,84 | 99,83 |
| No of labs without outliers | 19 | 19 | 20 | 18 | 18 | 18 |
| Repeatability standard deviation, s r (g/l) | 0,09 | 0,75 | 2,17 | 5,99 | 4,31 | 1,25 |
| Repeatability relative standard deviation, RSD r (%) | 0,81 | 3,07 | 2,1 | 1,71 | 1,35 | 1,25 |
| Repeatability limit, r (g/l) (r = 2,8 × s r ) | 0,25 | 2,1 | 6,07 | 16,76 | 12,06 | 3,49 |
| Reproducibility standard deviation, s R (g/l) | 0,79 | 0,92 | 4,18 | 9,94 | 16,11 | 4,63 |
| Reproducibility relative standard deviation, RSD R (%) | 7,31 | 3,76 | 4,05 | 2,84 | 5,04 | 4,64 |
| Reproducibility limit, R (g/l) (R = 2,8 × s R ) | 2,22 | 2,57 | 11,7 | 27,84 | 45,12 | 12,97 |
Table 3
Total Sugars
| ( Note: this data was calculated for total sugars, not invert sugar as defined in Section 8 above.) | |||||||
| a split level.] | |||||||
| Samples | Pastis | Ouzo | Aniseed-flavoured spirit drink | Cherry liqueur | Crème de Menthe | Crème de Cassis | Standard (220 g/l) |
|---|---|---|---|---|---|---|---|
| Mean value (g/l) | 10,86 | 29,2 19,7 a | 31,59 | 103,33 | 349,73 | 509,69 | 218,78 |
| No of Labs without outliers | 20 | 19 | 20 | 20 | 18 | 18 | 19 |
| Repeatability standard deviation, s r (g/l) | 0,13 | 0,75 | 0,77 | 2,17 | 5,89 | 5,59 | 2,71 |
| Repeatability relative standard deviation, RSD r (%) | 1,16 | 3,07 | 2,45 | 2,1 | 1,69 | 1,1 | 1,24 |
| Repeatability limit, r (g/l) (r = 2,8 × s r ) | 0,35 | 2,1 | 2,17 | 6,07 | 16,5 | 15,65 | 7,59 |
| Reproducibility standard deviation s R (g/l) | 0,79 | 0,92 | 1,51 | 4,18 | 9,98 | 14,81 | 8,53 |
| Reproducibility relative standard deviation, RSD R (%) | 7,25 | 3,76 | 4,79 | 4,04 | 2,85 | 2,91 | 3,9 |
| Reproducibility limit R (g/l) (R = 2,8 × s R ) | 2,21 | 2,57 | 4,24 | 11,7 | 27,94 | 41,48 | 23,89 |
IX. EGG YOLK. DETERMINATION OF EGG YOLK CONCENTRATION IN SPIRIT DRINKS — PHOTOMETRIC METHOD U.K.
1. Scope U.K.
This method is suitable for the determination of egg yolk concentration in the range of 40 to 250 g/l in egg liqueur and liqueur with egg.
2. Normative references U.K.
ISO 3696:1897 Water for analytical laboratory use — Specifications and test methods.
3. Principle U.K.
The ethanol-soluble phosphorus compounds found in egg yolk are extracted and assayed photometrically as a phosphorus molybdate complex.
4. Reagents and materials U.K.
4.1. Double-distilled water U.K.
4.2. Diatomaceous earth U.K.
4.3. Ethanol 96 % vol. (CAS 64-17-5) U.K.
4.4. 15 % magnesium acetate (CAS 16674-78-5) solution U.K.
4.5. 10 % sulphuric acid (CAS 7664-93-9) U.K.
4.6. 1 N sulphuric acid. U.K.
4.7. 0,16 g/l potassium dihydrogen phosphate (CAS 778-77-0), KH 2 PO 4 solution U.K.
4.8. Reagent for phosphate determination: U.K.
dissolve 20 g of ammonium molybdate (CAS 12054-85-2), (NH 4 ) 6 Mo 7 O 24 .4H 2 O in 400 ml water at 50 o C;
dissolve, in another vessel, 1 g of ammonium vanadate (CAS 7803-55-6), NH 4 VO 3 , in 300 ml hot water, allow to cool, then add 140 ml of concentrated nitric acid (CAS 7697-37-2). Combine the cooled solutions in a 1 000 ml volumetric flask and make up to the 1 000 ml mark.
5. Apparatus and equipment U.K.
5.1. 100 ml conical flask U.K.
5.2. Ultrasonic bath (or magnetic stirrer) U.K.
5.3. 100 ml volumetric flask U.K.
5.4. 20 o C water bath U.K.
5.5. Filter (Whatman No 4 or equivalent) U.K.
5.6. Porcelain (or platinum) crucible U.K.
5.7. Boiling water bath U.K.
5.8. Hot plate U.K.
5.9. Muffle furnace U.K.
5.10. 50 ml volumetric flask U.K.
5.11. 20 ml volumetric flask U.K.
5.12. Spectrophotometer set at 420 nm U.K.
5.13. 1 cm cuvette. U.K.
6. Samples U.K.
Samples are stored at room temperature prior to analysis.
7. Procedure U.K.
7.1. Sample preparation U.K.
7.1.1. Weigh 10 g of the sample into a 100 ml conical flask (5.1). U.K.
7.1.2. Add gradually 70 ml of ethanol (4.3) in small portions, swirling with each addition, and place in an ultrasonic bath (5.2) for 15 minutes (or stir the mixture with a magnetic stirrer (5.2) for 10 minutes at room temperature). U.K.
7.1.3. Transfer the contents of the flask to a 100 ml volumetric flask (5.3) with washings of ethanol (4.3). Adjust to the calibration mark with ethanol (4.3) and place the flasks in a 20 o C water bath (5.4). Adjust to the calibration mark at 20 o C. U.K.
7.1.4. Add a small amount of diatomaceous earth (4.2) and filter (5.5), discarding the first 20 ml. U.K.
7.1.5. Transfer 25 ml of the filtrate to a porcelain (or platinum) crucible (5.6). The filtrate must then be concentrated by gentle evaporation in a boiling water bath (5.7), with the addition of 5 ml of 15 % magnesium acetate solution (4.4). U.K.
7.1.6. Place the crucibles on a hot plate (5.8) and heat until just dry. U.K.
7.1.7. Ash the residue by heating to incandescence at 600 o C in a muffle furnace (5.9) until the ash is white, minimum of one and a half hours but can be left overnight. U.K.
7.1.8. Take up the ash with 10 ml of 10 % sulphuric acid (4.5) and transfer it with washings of distilled water (4.1) to a 50 ml volumetric flask (5.10), and fill to the mark at room temperature with distilled water (4.1). A 5 ml aliquot of this ash solution is to be used to prepare the sample solution of the photometric phosphate assay. U.K.
7.2. Photometric phosphate assay U.K.
7.2.1. Comparative solution U.K.
7.2.1.1. Place 10 ml of 10 % sulphuric acid (4.5) in a 50 ml volumetric flask (5.10) and fill to the mark with distilled water (4.1). U.K.
7.2.1.2. Add to a 5 ml aliquot of this solution (7.2.1.1), contained in a 20 ml volumetric flask (5.11), 1 ml of 1 N sulphuric acid (4.6) and 2 ml of the phosphate reagent (4.8) and make up to 20 ml with distilled water (4.1). U.K.
7.2.1.3. Stopper with a loosely inserted stopper, shake, and heat in a boiling water bath (5.7) for 10 minutes, then cool in a 20 o C water bath (5.4) for 20 minutes. U.K.
7.2.1.4. Fill a 1 cm cuvette (5.13) with this comparative solution. U.K.
7.2.2. Sample solution U.K.
7.2.2.1. Add to a 5 ml aliquot of the ash solution (7.1.8), contained in a 20 ml volumetric flask (5.11), 1 ml of 1 N sulphuric acid (4.6) and 2 ml of the phosphate reagent (4.8) and make up to 20 ml with distilled water (4.1). U.K.
7.2.2.2. Stopper with a loosely inserted stopper, shake, and heat in a boiling water bath (5.7) for 10 minutes, then cool in a 20 o C water bath (5.4) for 20 minutes. U.K.
7.2.2.3. The yellow solution that develops is immediately analysed spectrophotometrically (5.12) in a 1 cm cuvette (5.13) at 420 nm against the comparative solution (7.2.1.4). U.K.
7.2.3. Calibration curve U.K.
7.2.3.1. To construct the calibration curve, add 2 ml aliquots of the phosphate reagent (4.8) to 20 ml volumetric flasks (5.11) each containing 1 ml of 1 N sulphuric acid (4.6) and 0, 2, 4, 6, 8, and 10 ml of the potassium dihydrogen phosphate solution (4.7) respectively, and make up to the 20 ml mark with distilled water (4.1). U.K.
7.2.3.2. Stopper with a loosely inserted stopper, shake, and heat in a boiling water bath (5.7) for 10 minutes, then cool in a 20 o C water bath (5.4) for 20 minutes and analyse spectrophotometrically (5.12) in a 1 cm cuvette (5.13) at 420 nm against the comparative solution (7.2.1.4). U.K.
7.2.3.3. Construction of the calibration curve: U.K.
| dihydrogen phosphate solution (ml) | 0 | 2 | 4 | 6 | 8 | 10 |
| P 2 O 5 (mg) | 0 | 0,167 | 0,334 | 0,501 | 0,668 | 0,835 |
8. Expression of results U.K.
The egg yolk content in g/l is calculated from the following formula: 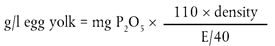
where:
conversion factor for total P 2 O 5 in g in 100 g of egg yolk
value established from the calibration curve
mass per unit volume (g/ml) of the egg-based liqueur at 20 o C
weight of the egg-based liqueur in g
dilution factor for a 5 ml aliquot of ash solution.
9. Method performance characteristics (precision) U.K.
Statistical results of the interlaboratory test:
the following table gives the values for egg yolk.
The following data were obtained from an international method performance study carried out to internationally agreed procedures.
Year of interlaboratory test: 1998 Number of laboratories: 24 Number of samples: 5 Analyte: Egg yolk Samples A B C D E Number of laboratories retained after eliminating outliers 19 20 22 20 22 Number of outliers (laboratories) 3 4 2 4 2 Number of accepted results 38 40 44 40 44 Mean value 147,3 241,1 227,4 51,9 (*)
72,8 (*)
191,1 Repeatability standard deviation (S r ) g/l 2,44 4,24 3,93 1,83 3,25 Repeatability relative standard deviation (RSD r ) (%) 1,7 1,8 1,8 2,9 1,7 Repeatability limit (r) g/l 6,8 11,9 11,0 5,1 9,1 Reproducibility standard deviation (S R ) g/l 5,01 6,06 6,66 3,42 6,87 Reproducibility relative standard deviation (RSD R ) (%) 3,4 2,5 2,9 5,5 3,6 Reproducibility limit (R) g/l 14,0 17,0 18,7 9,6 19,2 Sample types
AAdvocaat, blind duplicates
BAdvocaat, blind duplicates
CAdvocaat, blind duplicates
DAdvocaat (diluted), split levels (*)
EAdvocaat, blind duplicates]
[F3X. DETERMINATION OF THE FOLLOWING WOOD COMPOUNDS IN SPIRIT DRINKS BY HIGH PERFORMANCE LIQUID CHROMATOGRAPHY (HPLC): FURFURAL, 5-HYDROXYMETHYLFURFURAL, 5-METHYLFURFURAL, VANILLIN, SYRINGALDEHYDE, CONIFERALDEHYDE, SINAPALDEHYDE, GALLIC ACID, ELLAGIC ACID, VANILLIC ACID, SYRINGIC ACID AND SCOPOLETIN U.K.
1. Scope U.K.
The method pertains to the determination of furfural, 5-hydroxymethylfurfural, 5-methylfurfural, vanillin, syringaldehyde, coniferaldehyde, sinapaldehyde, gallic acid, ellagic acid, vanillic acid, syringic acid and scopoletin, by high-performance liquid chromatography.
2. Normative reference U.K.
Analytical method recognised by the General Assembly of the International Organisation of Vine and Wine (OIV) and published by OIV under the reference OIV-MA-BS-16: R2009 .
3. Principle U.K.
Determination by high-performance liquid chromatography (HPLC), with detection by ultraviolet spectrophotometry at several wavelengths and by spectrofluorimetry.
4. Reagents U.K.
The reagents must be of analytical quality. The water used must be distilled water or water of at least equivalent purity. It is preferable to use microfiltered water with a resistivity of 18,2 M Ω.cm.
4.1. 96 % vol. alcohol. U.K.
4.2. HPLC-quality methanol (Solvent B). U.K.
4.3. Acetic acid diluted to 0,5 % vol. (Solvent A). U.K.
4.4. Mobile phases: (given as an example only). U.K.
Solvent A (0,5 % acetic acid) and solvent B (pure methanol). Filter through a membrane (porosity 0,45 μm). Degas in an ultrasonic bath, if necessary.
4.5. Reference standards of 99 % minimum purity: furfural, 5-hydroxymethyl furfural, 5-methylfurfural, vanillin, syringaldehyde, coniferaldehyde, sinapaldehyde, gallic acid, ellagic acid, vanillic acid, syringic acid and scopoletin. U.K.
4.6. Reference solution: the standard substances are dissolved in a 50 % vol. aqueous-alcoholic solution. The final concentrations in the reference solution should be of the order of: U.K.
furfural: 5 mg/l; 5-hydroxymethyl furfural: 10 mg/l; 5-methylfurfural 2 mg/l; vanillin: 5 mg/l; syringaldehyde: 10 mg/l; coniferaldehyde: 5 mg/l; sinapaldehyde: 5 mg/l; gallic acid: 10 mg/l; ellagic acid: 10 mg/l; vanillic acid: 5 mg/l; syringic acid: 5 mg/l; scopoletin: 0,5 mg/l.
5. Apparatus U.K.
Standard laboratory apparatus
5.1. A high-performance liquid chromatograph capable of functioning in binary gradient mode and equipped with: U.K.
A spectrophotometric detector capable of measuring at wavelengths from 260 to 340 nm. It is however preferable to work with a multiple wavelength detector with a diode array or similar, in order to confirm the purity of the peaks.
A spectrofluorimetric detector — excitation wavelength: 354 nm, emission wavelength: 446 nm (for the trace determination of scopoletin; which is also detectable at 313 nm by spectrophotometry).
An injection device capable of introducing 10 or 20 μl (for example) of the test sample.
A high-performance liquid chromatography column, RP C18 type, 5 μm maximum particle size.
5.2. Syringes for HPLC. U.K.
5.3. Device for membrane-filtration of small volumes. U.K.
5.4. Integrator-computer or recorder with performance compatible with the entire apparatus, and in particular, it must have several acquisition channels. U.K.
6. Procedure U.K.
6.1. Preparation of the solution to be injected U.K.
The reference solution and the spirit drink are filtered, if necessary, through a membrane with a maximum pore diameter of 0,45 μm.
6.2. Chromatographic operating conditions: carry out the analysis at ambient temperature by means of the equipment described in (5.1) and using the mobile phases (4.4) with a flow of approximately 0,6 ml per minute following the gradient below (given as an example only) U.K.
Time: 0 min 50 min 70 min 90 min
solvent A (water-acid): 100 % 60 % 100 % 100 %
solvent B (methanol): 0 % 40 % 0 % 0 %
Note that in certain cases this gradient should be modified to avoid co-elutions.
6.3. Determination U.K.
6.3.1. Inject the reference standards separately, then mixed. U.K.
Adapt the operating conditions so that the resolution factors of the peaks of all the compounds are equal to at least 1.
6.3.2. Inject the sample as prepared in 6.1. U.K.
6.3.3. Measure the area of the peaks in the reference solution and the spirit drink and calculate the concentrations. U.K.
7. Expression of results U.K.
Express the concentration of each constituent in mg/l.
8. Performance characteristics of the method (precision) U.K.
The following data were obtained in 2009 from an international method-performance study on a variety of spirit drinks, carried out following internationally-agreed procedures (1) (2).
8.1. Furfural U.K.
| Analyte | Furfural | |||||
|---|---|---|---|---|---|---|
| Samples | Whisky | Brandy | Rum | Cognac 1 | Bourbon | Cognac 2 |
| No of laboratories participating | 15 | 15 | 15 | 15 | 15 | 15 |
| No of results accepted (laboratories) | 14 | 12 | 13 | 14 | 13 | 13 |
| Mean value (mg/l) | 2,9 | 1,2 | 1,7 | 10,6 | 15,3 | 13,9 |
| Repeatability standard deviation, s r (mg/l) | 0,04 | 0,05 | 0,04 | 0,18 | 0,23 | 0,20 |
| Repeatability relative standard deviation, RSD r (%) | 1,4 | 4,5 | 2,3 | 1,7 | 1,5 | 1,5 |
| Repeatability limit, r (mg/l) (r = 2,8 × s r ) | 0,1 | 0,2 | 0,1 | 0,5 | 0,6 | 0,6 |
| Reproducibility standard deviation, s R (mg/l) | 0,24 | 0,18 | 0,09 | 1,4 | 0,49 | 0,69 |
| Reproducibility relative standard deviation, RSD R (%) | 8 | 15 | 5 | 13 | 3 | 5 |
| Reproducibility limit, R (g/l) (R = 2,8 × s R ) | 0,7 | 0,5 | 0,3 | 3,8 | 1,4 | 1,9 |
8.2. 5-Hydroxymethylfurfural U.K.
| Analyte | 5-Hydroxymethylfurfural | |||||
|---|---|---|---|---|---|---|
| Samples | Whisky | Brandy | Rum | Cognac 1 | Bourbon | Cognac 2 |
| No of laboratories participating | 16 | 16 | 16 | 16 | 16 | 16 |
| No of results accepted (laboratories) | 14 | 14 | 14 | 14 | 14 | 14 |
| Mean value (mg/l) | 5,0 | 11,1 | 9,4 | 33,7 | 5,8 | 17,5 |
| Repeatability standard deviation, s r (mg/l) | 0,09 | 0,09 | 0,09 | 0,42 | 0,07 | 0,13 |
| Repeatability relative standard deviation, RSD r (%) | 1,7 | 0,8 | 1,0 | 1,3 | 1,2 | 0,8 |
| Repeatability limit, r (mg/l) (r = 2,8 × s r ) | 0,2 | 0,3 | 0,3 | 1,2 | 0,2 | 0,4 |
| Reproducibility standard deviation, s R (mg/l) | 0,39 | 1,01 | 0,50 | 4,5 | 0,4 | 1,6 |
| Reproducibility relative standard deviation, RSD R (%) | 8 | 9 | 5 | 13 | 7 | 9 |
| Reproducibility limit, R (g/l) (R = 2,8 × s R ) | 1,1 | 2,8 | 1,4 | 12,5 | 1,1 | 4,6 |
8.3. 5-Methylfurfural U.K.
| Analyte | 5-Methylfurfural | |||||
|---|---|---|---|---|---|---|
| Samples | Whisky | Brandy | Rum | Cognac 1 | Bourbon | Cognac 2 |
| No of laboratories participating | 11 | 11 | 11 | 11 | 11 | 11 |
| No of results accepted (laboratories) | 11 | 11 | 8 | 11 | 10 | 11 |
| Mean value (mg/l) | 0,1 | 0,2 | 0,1 | 0,5 | 1,7 | 0,8 |
| Repeatability standard deviation, s r (mg/l) | 0,01 | 0,01 | 0,02 | 0,02 | 0,03 | 0,07 |
| Repeatability relative standard deviation, RSD r (%) | 10,7 | 6,1 | 13,6 | 4,7 | 2,0 | 10,0 |
| Repeatability limit, r (mg/l) (r = 2,8 × s r ) | 0,0 | 0,0 | 0,1 | 0,1 | 0,1 | 0,2 |
| Reproducibility standard deviation, s R (mg/l) | 0,03 | 0,04 | 0,03 | 0,18 | 0,20 | 0,26 |
| Reproducibility relative standard deviation, RSD R (%) | 35 | 18 | 22 | 39 | 12 | 35 |
| Reproducibility limit, R (g/l) (R = 2,8 × s R ) | 0,1 | 0,1 | 0,1 | 0,5 | 0,6 | 0,7 |
8.4. Vanillin U.K.
| Analyte | Vanillin | |||||
|---|---|---|---|---|---|---|
| Samples | Whisky | Brandy | Rum | Cognac 1 | Bourbon | Cognac 2 |
| No of laboratories participating | 16 | 15 | 16 | 16 | 16 | 16 |
| No of results accepted (laboratories) | 16 | 15 | 16 | 16 | 16 | 16 |
| Mean value (mg/l) | 0,5 | 0,2 | 1,2 | 1,2 | 3,2 | 3,9 |
| Repeatability standard deviation, s r (mg/l) | 0,03 | 0,02 | 0,06 | 0,11 | 0,11 | 0,09 |
| Repeatability relative standard deviation, RSD r (%) | 6,8 | 9,6 | 4,6 | 8,9 | 3,5 | 2,3 |
| Repeatability limit, r (mg/l) (r = 2,8 × s r ) | 0,1 | 0,1 | 0,2 | 0,3 | 0,3 | 0,3 |
| Reproducibility standard deviation, s R (mg/l) | 0,09 | 0,06 | 0,18 | 0,27 | 0,41 | 0,62 |
| Reproducibility relative standard deviation, RSD R (%) | 19 | 25 | 15 | 22 | 13 | 16 |
| Reproducibility limit, R (g/l) (R = 2,8 × s R ) | 0,3 | 0,2 | 0,5 | 0,8 | 1,2 | 1,7 |
8.5. Syringaldehyde U.K.
| Analyte | Syringaldehyde | |||||
|---|---|---|---|---|---|---|
| Samples | Whisky | Brandy | Rum | Cognac 1 | Bourbon | Cognac 2 |
| No of laboratories participating | 16 | 15 | 16 | 16 | 16 | 16 |
| No of results accepted (laboratories) | 13 | 13 | 13 | 12 | 14 | 13 |
| Mean value (mg/l) | 1,0 | 0,2 | 4,8 | 3,2 | 10,5 | 9,7 |
| Repeatability standard deviation, s r (mg/l) | 0,03 | 0,02 | 0,04 | 0,08 | 0,10 | 0,09 |
| Repeatability relative standard deviation, RSD r (%) | 2,6 | 8,1 | 0,8 | 2,6 | 0,9 | 0,9 |
| Repeatability limit, r (mg/l) (r = 2,8 × s r ) | 0,1 | 0,1 | 0,1 | 0,2 | 0,3 | 0,3 |
| Reproducibility standard deviation, s R (mg/l) | 0,08 | 0,07 | 0,23 | 0,19 | 0,39 | 0,43 |
| Reproducibility relative standard deviation, RSD R (%) | 8 | 33 | 5 | 6 | 4 | 4 |
| Reproducibility limit, R (g/l) (R = 2,8 × s R ) | 0,2 | 0,2 | 0,7 | 0,5 | 1,1 | 1,2 |
8.6. Coniferaldehyde U.K.
| Analyte | Coniferaldehyde | |||||
|---|---|---|---|---|---|---|
| Samples | Whisky | Brandy | Rum | Cognac 1 | Bourbon | Cognac 2 |
| No of laboratories participating | 13 | 12 | 13 | 12 | 13 | 13 |
| No of results accepted (laboratories) | 12 | 12 | 13 | 12 | 13 | 13 |
| Mean value (mg/l) | 0,2 | 0,2 | 0,6 | 0,8 | 4,6 | 1,3 |
| Repeatability standard deviation, s r (mg/l) | 0,02 | 0,02 | 0,03 | 0,03 | 0,09 | 0,06 |
| Repeatability relative standard deviation, RSD r (%) | 9,2 | 9,8 | 4,6 | 4,3 | 1,9 | 4,5 |
| Repeatability limit, r (mg/l) (r = 2,8 × s r ) | 0,04 | 0,04 | 0,07 | 0,09 | 0,24 | 0,16 |
| Reproducibility standard deviation, s R (mg/l) | 0,04 | 0,04 | 0,11 | 0,18 | 0,38 | 0,25 |
| Reproducibility relative standard deviation, RSD R (%) | 23 | 27 | 21 | 23 | 8 | 19 |
| Reproducibility limit, R (g/l) (R = 2,8 × s R ) | 0,1 | 0,1 | 0,3 | 0,5 | 1,1 | 0,7 |
8.7. Sinapaldehyde U.K.
| Analyte | Sinapaldehyde | |||||
|---|---|---|---|---|---|---|
| Samples | Whisky | Brandy | Rum | Cognac 1 | Bourbon | Cognac 2 |
| No of laboratories participating | 14 | 14 | 14 | 14 | 15 | 14 |
| No of results accepted (laboratories) | 14 | 13 | 12 | 13 | 13 | 12 |
| Mean value (mg/l) | 0,3 | 0,2 | 0,2 | 1,6 | 8,3 | 0,3 |
| Repeatability standard deviation, s r (mg/l) | 0,02 | 0,01 | 0,02 | 0,06 | 0,14 | 0,03 |
| Repeatability relative standard deviation, RSD r (%) | 7,5 | 4,6 | 11,2 | 3,7 | 1,6 | 11,4 |
| Repeatability limit, r (mg/l) (r = 2,8 × s r ) | 0,06 | 0,03 | 0,06 | 0,17 | 0,38 | 0,08 |
| Reproducibility standard deviation, s R (mg/l) | 0,09 | 0,05 | 0,08 | 0,20 | 0,81 | 0,18 |
| Reproducibility relative standard deviation, RSD R (%) | 31 | 27 | 46 | 13 | 10 | 73 |
| Reproducibility limit, R (g/l) (R = 2,8 × s R ) | 0,2 | 0,2 | 0,2 | 0,6 | 2,3 | 0,5 |
8.8. Gallic acid U.K.
| Analyte | Gallic acid | |||||
|---|---|---|---|---|---|---|
| Sample | Whisky | Brandy | Rum | Cognac 1 | Bourbon | Cognac 2 |
| No of laboratories participating | 16 | 15 | 16 | 16 | 16 | 16 |
| No of results accepted (laboratories) | 15 | 14 | 16 | 16 | 16 | 16 |
| Mean value (mg/l) | 1,2 | 0,4 | 2,0 | 6,1 | 7,3 | 21,8 |
| Repeatability standard deviation, s r (mg/l) | 0,07 | 0,04 | 0,06 | 0,18 | 0,18 | 0,60 |
| Repeatability relative standard deviation, RSD r (%) | 6,1 | 8,1 | 2,9 | 3,0 | 2,4 | 2,8 |
| Repeatability limit, r (mg/l) (r = 2,8 × s r ) | 0,2 | 0,1 | 0,2 | 0,5 | 0,5 | 1,7 |
| Reproducibility standard deviation, s R (mg/l) | 0,43 | 0,20 | 0,62 | 3,3 | 2,2 | 7,7 |
| Reproducibility relative standard deviation, RSD R (%) | 36 | 47 | 31 | 53 | 30 | 35 |
| Reproducibility limit, R (g/l) (R = 2,8 × s R ) | 1,2 | 0,6 | 1,7 | 9,1 | 6,2 | 21,7 |
8.9. Ellagic acid U.K.
| Analyte | Ellagic acid | |||||
|---|---|---|---|---|---|---|
| Samples | Whisky | Brandy | Rum | Cognac 1 | Bourbon | Cognac 2 |
| No of laboratories participating | 7 | 7 | 7 | 7 | 7 | 7 |
| No of results accepted (laboratories) | 7 | 7 | 7 | 7 | 7 | 6 |
| Mean value (mg/l) | 3,2 | 1,0 | 9,5 | 13 | 13 | 36 |
| Repeatability standard deviation, s r (mg/l) | 0,20 | 0,16 | 0,30 | 0,41 | 0,95 | 0,34 |
| Repeatability relative standard deviation, RSD r (%) | 6,3 | 16 | 3,2 | 3,2 | 7,4 | 1,0 |
| Repeatability limit, r (mg/l) (r = 2,8 × s r ) | 0,6 | 0,4 | 0,9 | 1,1 | 2,7 | 1,0 |
| Reproducibility standard deviation, s R (mg/l) | 1,41 | 0,42 | 4,0 | 5,0 | 4,9 | 14 |
| Reproducibility relative standard deviation, RSD R (%) | 44 | 43 | 42 | 39 | 39 | 40 |
| Reproducibility limit, R (g/l) (R = 2,8 × s R ) | 4,0 | 1,2 | 11 | 14 | 14 | 40 |
8.10. Vanillic acid U.K.
| Analyte | Vanillic acid | |||||
|---|---|---|---|---|---|---|
| Samples | Whisky | Brandy | Rum | Cognac 1 | Bourbon | Cognac 2 |
| No of laboratories participating | 15 | 15 | 15 | 15 | 15 | 15 |
| No of results accepted (laboratories) | 12 | 11 | 14 | 14 | 15 | 14 |
| Mean value (mg/l) | 0,2 | 0,2 | 1,5 | 0,8 | 2,4 | 2,7 |
| Repeatability standard deviation, s r (mg/l) | 0,03 | 0,04 | 0,03 | 0,10 | 0,13 | 0,21 |
| Repeatability relative standard deviation, RSD r (%) | 14,2 | 16,5 | 2,3 | 12,6 | 5,3 | 7,7 |
| Repeatability limit, r (mg/l) (r = 2,8 × s r ) | 0,1 | 0,1 | 0,1 | 0,3 | 0,4 | 0,6 |
| Reproducibility standard deviation, s R (mg/l) | 0,06 | 0,05 | 0,51 | 0,2 | 1,22 | 0,70 |
| Reproducibility relative standard deviation, RSD R (%) | 28 | 20 | 35 | 31 | 51 | 26 |
| Reproducibility limit, R (g/l) (R = 2,8 × s R ) | 0,2 | 0,1 | 1,4 | 0,7 | 3,4 | 2,0 |
8.11. Syringic acid U.K.
| Analyte | Syringic acid | |||||
|---|---|---|---|---|---|---|
| Samples | Whisky | Brandy | Rum | Cognac 1 | Bourbon | Cognac 2 |
| No of laboratories participating | 16 | 15 | 16 | 16 | 16 | 16 |
| No of results accepted (laboratories) | 16 | 15 | 15 | 15 | 16 | 15 |
| Mean value (mg/l) | 0,4 | 0,2 | 2,5 | 1,4 | 3,4 | 4,8 |
| Repeatability standard deviation, s r (mg/l) | 0,03 | 0,02 | 0,06 | 0,13 | 0,08 | 0,11 |
| Repeatability relative standard deviation, RSD r (%) | 6,7 | 12,6 | 2,3 | 9,0 | 2,3 | 2,3 |
| Repeatability limit, r (mg/l) (r = 2,8 × s r ) | 0,1 | 0,1 | 0,2 | 0,4 | 0,2 | 0,3 |
| Reproducibility standard deviation, s R (mg/l) | 0,08 | 0,05 | 0,29 | 0,26 | 0,43 | 0,67 |
| Reproducibility relative standard deviation, RSD R (%) | 19 | 29 | 11 | 18 | 13 | 14 |
| Reproducibility limit, R (g/l) (R = 2,8 × s R ) | 0,2 | 0,1 | 0,8 | 0,7 | 1,2 | 1,9 |
8.12. Scopoletin U.K.
| Analyte | Scopoletin | |||||
|---|---|---|---|---|---|---|
| Samples | Whisky | Brandy | Rum | Cognac 1 | Bourbon | Cognac 2 |
| No of laboratories participating | 10 | 10 | 10 | 10 | 10 | 10 |
| No of results accepted (laboratories) | 9 | 8 | 9 | 8 | 8 | 8 |
| Mean value (mg/l) | 0,09 | 0,04 | 0,11 | 0,04 | 0,65 | 0,15 |
| Repeatability standard deviation, s r (mg/l) | 0,0024 | 0,0008 | 0,0018 | 0,0014 | 0,0054 | 0,0040 |
| Repeatability relative standard deviation, RSD r (%) | 2,6 | 2,2 | 1,6 | 3,3 | 0,8 | 2,7 |
| Repeatability limit, r (mg/l) (r = 2,8 × s r ) | 0,007 | 0,002 | 0,005 | 0,004 | 0,015 | 0,011 |
| Reproducibility standard deviation, s R (mg/l) | 0,01 | 0,01 | 0,03 | 0,01 | 0,09 | 0,02 |
| Reproducibility relative standard deviation, RSD R (%) | 15 | 16 | 23 | 17 | 15 | 15 |
| Reproducibility limit, R (g/l) (R = 2,8 × s R ) | 0,04 | 0,02 | 0,07 | 0,02 | 0,26 | 0,06 |
(1) ‘Protocol for the design, conduct and interpretation of method-performance studies’, Horwitz, W. (1995) Pure and Applied Chemistry , 67, 332-343. U.K.
(2) Horwitz, W. (1982) Analytical Chemistry , 54, 67A-76A.] U.K.
Options/Help
Print Options
PrintThe Whole Regulation
PrintThe Annexes only
You have chosen to open the Whole Regulation
The Whole Regulation you have selected contains over 200 provisions and might take some time to download. You may also experience some issues with your browser, such as an alert box that a script is taking a long time to run.
Would you like to continue?
You have chosen to open Schedules only
Y Rhestrau you have selected contains over 200 provisions and might take some time to download. You may also experience some issues with your browser, such as an alert box that a script is taking a long time to run.
Would you like to continue?
Mae deddfwriaeth ar gael mewn fersiynau gwahanol:
Y Diweddaraf sydd Ar Gael (diwygiedig):Y fersiwn ddiweddaraf sydd ar gael o’r ddeddfwriaeth yn cynnwys newidiadau a wnaed gan ddeddfwriaeth ddilynol ac wedi eu gweithredu gan ein tîm golygyddol. Gellir gweld y newidiadau nad ydym wedi eu gweithredu i’r testun eto yn yr ardal ‘Newidiadau i Ddeddfwriaeth’.
Gwreiddiol (Fel y’i mabwysiadwyd gan yr UE): Mae'r wreiddiol version of the legislation as it stood when it was first adopted in the EU. No changes have been applied to the text.
Pwynt Penodol mewn Amser: This becomes available after navigating to view revised legislation as it stood at a certain point in time via Advanced Features > Show Timeline of Changes or via a point in time advanced search.
Gweler y wybodaeth ychwanegol ochr yn ochr â’r cynnwys
Rhychwant ddaearyddol: Indicates the geographical area that this provision applies to. For further information see ‘Frequently Asked Questions’.
Dangos Llinell Amser Newidiadau: See how this legislation has or could change over time. Turning this feature on will show extra navigation options to go to these specific points in time. Return to the latest available version by using the controls above in the What Version box.
Rhagor o Adnoddau
Gallwch wneud defnydd o ddogfennau atodol hanfodol a gwybodaeth ar gyfer yr eitem ddeddfwriaeth o’r tab hwn. Yn ddibynnol ar yr eitem ddeddfwriaeth sydd i’w gweld, gallai hyn gynnwys:
- y PDF print gwreiddiol y fel adopted version that was used for the EU Official Journal
- rhestr o newidiadau a wnaed gan a/neu yn effeithio ar yr eitem hon o ddeddfwriaeth
- pob fformat o’r holl ddogfennau cysylltiedig
- slipiau cywiro
- dolenni i ddeddfwriaeth gysylltiedig ac adnoddau gwybodaeth eraill
Llinell Amser Newidiadau
Mae’r llinell amser yma yn dangos y fersiynau gwahanol a gymerwyd o EUR-Lex yn ogystal ag unrhyw fersiynau dilynol a grëwyd ar ôl y diwrnod ymadael o ganlyniad i newidiadau a wnaed gan ddeddfwriaeth y Deyrnas Unedig.
Cymerir dyddiadau fersiynau’r UE o ddyddiadau’r dogfennau ar EUR-Lex ac efallai na fyddant yn cyfateb â’r adeg pan ddaeth y newidiadau i rym ar gyfer y ddogfen.
Ar gyfer unrhyw fersiynau a grëwyd ar ôl y diwrnod ymadael o ganlyniad i newidiadau a wnaed gan ddeddfwriaeth y Deyrnas Unedig, bydd y dyddiad yn cyd-fynd â’r dyddiad cynharaf y daeth y newid (e.e. ychwanegiad, diddymiad neu gyfnewidiad) a weithredwyd i rym. Am ragor o wybodaeth gweler ein canllaw i ddeddfwriaeth ddiwygiedig ar Ddeall Deddfwriaeth.
Rhagor o Adnoddau
Defnyddiwch y ddewislen hon i agor dogfennau hanfodol sy’n cyd-fynd â’r ddeddfwriaeth a gwybodaeth am yr eitem hon o ddeddfwriaeth. Gan ddibynnu ar yr eitem o ddeddfwriaeth sy’n cael ei gweld gall hyn gynnwys:
- y PDF print gwreiddiol y fel adopted fersiwn a ddefnyddiwyd am y copi print
- slipiau cywiro
liciwch ‘Gweld Mwy’ neu ddewis ‘Rhagor o Adnoddau’ am wybodaeth ychwanegol gan gynnwys
- rhestr o newidiadau a wnaed gan a/neu yn effeithio ar yr eitem hon o ddeddfwriaeth
- manylion rhoi grym a newid cyffredinol
- pob fformat o’r holl ddogfennau cysylltiedig
- dolenni i ddeddfwriaeth gysylltiedig ac adnoddau gwybodaeth eraill