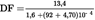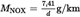[F1ANNEX III U.K. TYPE I TEST (Verifying the average emission of tailpipe emissions after a cold start)
Textual Amendments
Appendix 8 CALCULATION OF THE EMISSION OF POLLUTANTS
1. GENERAL U.K.
1.1. Emissions of gaseous pollutants are calculated by means of the following equation: U.K.
( 1 )
where:
=
mass emission of the pollutant i in grams per kilometre,
=
volume of the diluted exhaust gas expressed in litres per test and corrected to standard conditions (273,2 K and 101,33 kPa),
=
density of the pollutant i in grams per litre at normal temperature and pressure (273,2 K and 101,33 kPa),
=
humidity correction factor used for the calculation of the mass emissions of oxides of nitrogen (there is no humidity correction for HC and CO),
=
concentration of the pollutant i in the diluted exhaust gas expressed in ppm and corrected by the amount of the pollutant i contained in the dilution air,
=
actual distance corresponding to the operating cycle in km.
1.2. Volume determination U.K.
1.2.1. Calculation of the volume when a variable dilution device with constant flow control by orifice or venturi is used. Record continuously the parameters showing the volumetric flow, and calculate the total volume for the duration of the test. U.K.
1.2.2. Calculation of volume when a positive displacement pump is used. The volume of diluted exhaust gas in systems comprising a positive displacement pump is calculated with the following formula:  U.K.
U.K.
where:
=
volume of the diluted exhaust gas expressed in litres per test (prior to correction),
=
volume of gas delivered by the positive displacement pump on testing conditions in litres per revolution,
=
number of revolutions per test.
1.2.3. Correction of the diluted exhaust-gas volume to standard conditions. The diluted exhaust-gas volume is corrected by means of the following formula: U.K.
( 2 )
in which:
( 3 )
where:
=
barometric pressure in the test room in kPa,
=
vacuum at the inlet to the positive displacement pump in kPa relative to the ambient barometric pressure,
=
average temperature of the diluted exhaust gas entering the positive displacement pump during the test (K).
1.3. Calculation of the corrected concentration of pollutants in the sampling bag U.K.
( 4 )
where:
=
concentration of the pollutant i in the diluted exhaust gas, expressed in ppm and corrected by the amount of i contained in the dilution air,
=
measured concentration of pollutant i in the diluted exhaust gas, expressed in ppm,
=
measured concentration of pollutant i in the air used for dilution, expressed in ppm,
=
dilution factor.
[F2The dilution factor is calculated as follows: 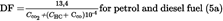
Textual Amendments
in this equation:
=
concentration of CO 2 in the diluted exhaust gas contained in the sampling bag, expressed in % volume,
=
concentration of HC in the diluted exhaust gas contained in the sampling bag, expressed in ppm carbon equivalent,
=
concentration of CO in the diluted exhaust gas contained in the sampling bag, expressed in ppm.
1.4. Determination of the NO humidity correction factor U.K.
In order to correct the influence of humidity on the results of oxides of nitrogen, the following calculations are applied:
( 6 )
where:
=
absolute humidity expressed in grams of water per kilogram of dry air,
=
relative humidity of the ambient air expressed as a percentage,
=
saturation vapour pressure at ambient temperature expressed in kPa,
=
atmospheric pressure in the room, expressed in kPa.
1.5. Example U.K.
1.5.1. Data U.K.
1.5.1.1. Ambient conditions: U.K.
ambient temperature: 23 °C = 296,2 K,
barometric pressure: P B = 101,33 kPa,
relative humidity: R a = 60 %,
[F3saturation vapour pressure: P d = 2,81 kPa of H 2 O at 23 °C.]
Textual Amendments
1.5.1.2. Volume measured and reduced to standard conditions (paragraph 1) U.K.
V = 51,961 m 3
1.5.1.3. Analyzer readings: U.K.
| a In ppm carbon equivalent. | ||
| Diluted exhaust | Dilution-air | |
|---|---|---|
| HC a | 92 ppm | 3,0 ppm |
| CO | 470 ppm | 0 ppm |
| NO x | 70 ppm | 0 ppm |
| CO 2 | 1,6 % vol | 0,03 % vol |
1.5.2. Calculation U.K.
[F31.5.2.1. Humidity correction factor (K H ) (see formula (6)) U.K.
H = 10,5092
k H = 0,9934]
1.5.2.2. Dilution factor (DF) (see formula (5)) U.K.
DF = 8,091
1.5.2.3. Calculation of the corrected concentration of pollutants in the sampling bag: U.K.
HC, mass emissions (see formulae (4) and (1))
C = 89,371
[F2Q HC = 0.619 in the case of petrol or diesel
Q HC = 0.649 in the case of LPG
Q HC = 0.714 in the case of NG]
CO, mass emissions (see formula (1))
Q CO = 1,25
NO x mass emissions (see formula (1))
Q NO x = 2,05
[F3