Please note that the date you requested in the address for this web page is not an actual date upon which a change occurred to this item of legislation. You are being shown the legislation from , which is the first date before then upon which a change was made.
[X1ANNEX U.K.
Editorial Information
CHAPTER I U.K. LEGAL UNITS OF MEASUREMENT REFERRED TO IN ARTICLE 1 (a)
1. SI UNITS AND THEIR DECIMAL MULTIPLES AND SUBMULTIPLES U.K.
1.1. SI base units U.K.
| Quantity | Unit | |
|---|---|---|
| Name | Symbol | |
| Length | metre | m |
| Mass | kilogram | kg |
| Time | second | s |
| Electric current | ampere | A |
| Thermodynamic temperature | kelvin | K |
| Amount of substance | mole | mol |
| Luminous intensity | candela | cd |
Definitions of SI base units: U.K.
[F1Unit of length U.K.
A metre is the length of the path travelled in a vacuum by light during 1/ 299 792 458 seconds.
(Seventeenth CGPM (1983), Resolution 1).]
Textual Amendments
Unit of mass U.K.
The kilogram is the unit of mass; it is equal to the mass of the international prototype of the kilogram.
(Third CGPM (1901), page 70 of the conference report).
Unit of time U.K.
The second is the duration of 9 192 631 770 periods of the radiation corresponding to the transition between the two hyperfine levels of the ground state of the caesium-133 atom.
(Thirteenth CGPM (1967), resolution 1).
Unit of electric current U.K.
The ampere is that constant current, which if maintained in two straight parallel conductors of infinite length, of negligible circular cross-section, and placed one metre apart in vacuum, would produce between those conductors a force equal to 2 × 10 -7 newton per metre of length.
(CIPM (1946), resolution 2, approved by the ninth CGPM (1948)).
[F2Unit of thermodynamic temperature U.K.
The kelvin, unit of thermodynamic temperature, is the fraction 1/273,16 of the thermodynamic temperature of the triple point of water.
This definition refers to water having the isotopic composition defined by the following amount-of-substance ratios: 0,00015576 mole of 2 H per mole of 1 H, 0,0003799 mole of 17 O per mole of 16 O and 0,0020052 mole of 18 O per mole of 16 O.
(Thirteenth CGPM (1967), resolution 4 and Twenty-third CGPM (2007), resolution 10)]
Textual Amendments
Unit of amount of substance U.K.
The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0·012 kilogram of carbon 12.
When the mole is used, the elementary entities must be specified and may be atoms, molecules, ions, electrons, other particles, or specified groups of such particles.
(Fourteenth CGPM (1971), resolution 3).
Unit of luminous intensity U.K.
The candela is the luminous intensity, in a given direction, of a source that emits monochromatic radiation of frequency of 540 × 10 12 hertz and that has a radiant intensity in that direction of (1/683) watt per steradian.
(Sixteenth CGPM (1979), resolution 3).
1.1.1. [F2Special name and symbol of the SI derived unit of temperature for expressing Celsius temperature] U.K.
| Quantity | Unit | |
|---|---|---|
| Name | Symbol | |
| Celsius temperature | degree Celsius | °C |
[F3Celsius temperature [X2t] is defined as the difference [X2t = T - T 0] between the two thermodynamic temperatures [X2T] and [X2T 0 where T 0] = 273,15 K. An interval or difference of temperature may be expressed either in kelvins or in degrees Celsius. The unit ‘ degree Celsius ’ is equal to the unit ‘ kelvin ’ .]
Editorial Information
X2 Substituted by Corrigendum to Directive 1999/103/EC of the European Parliament and of the Council of 24 January 2000 amending Council Directive 80/181/EEC on the approximation of the laws of the Member States relating to units of measurement (Official Journal of the European Communities L 34 of 9 February 2000).
Textual Amendments
[F21.2. SI derived units] U.K.
F41.2.1.SI supplementary unitsU.K.
| [ F4 |
F4. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .]
F4Definitions of SI supplementary units:U.K.
F4Unit of plane angleU.K.
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
F4Unit of solid angleU.K.
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
Textual Amendments
[F21.2.2. General rule for SI derived units U.K.
Units derived coherently from SI base units are given as algebraic expressions in the form of products of powers of the SI base units with a numerical factor equal to 1.
1.2.3. SI derived units with special names and symbols U.K.
| a Special names for the unit of power: the name volt–ampere (symbol ‘ VA ’ ) when it is used to express the apparent power of alternating electric current, and var (symbol ‘ var ’ ) when it is used to express reactive electric power. The ‘ var ’ is not included in GCPM resolutions. | ||||
| Quantity | Unit | Expression | ||
|---|---|---|---|---|
| Name | Symbol | In terms of other SI units | In terms of SI base units | |
| Plane angle | radian | rad | m · m –1 | |
| Solid angle | steradian | sr | m 2 · m –2 | |
| Frequency | hertz | Hz | s –1 | |
| Force | newton | N | m · kg · s –2 | |
| Pressure, stress | pascal | Pa | N · m –2 | m –1 · kg · s –2 |
| Energy, work; quantity of heat | joule | J | N · m | m 2 · kg · s –2 |
| Power a , radiant flux | watt | W | J · s –1 | m 2 · kg · s –3 |
| Quantity of electricity, electric charge | coulomb | C | s · A | |
| Electric potential, potential difference, electromotive force | volt | V | W · A –1 | m 2 · kg · s –3 · A –1 |
| Electric resistance | ohm | Ω | V · A –1 | m 2 · kg · s –3 · A –2 |
| Conductance | siemens | S | A · V –1 | m –2 · kg –1 · s 3 · A 2 |
| Capacitance | farad | F | C · V –1 | m –2 · kg –1 · s 4 · A 2 |
| Magnetic flux | weber | Wb | V · s | m 2 · kg · s –2 · A –1 |
| Magnetic flux density | tesla | T | Wb · m –2 | kg · s –2 · A –1 |
| Inductance | henry | H | Wb · A –1 | m 2 · kg · s –2 · A –2 |
| Luminous flux | lumen | lm | cd · sr | cd |
| Illuminance | lux | lx | lm · m –2 | m –2 · cd |
| Activity (of a radionuclide) | becquerel | Bq | s –1 | |
| Absorbed dose, specific energy imparted, kerma, absorbed dose index | gray | Gy | J · kg –1 | m 2 · s –2 |
| Dose equivalent | sievert | Sv | J · kg –1 | m 2 · s –2 |
| Catalytic activity | katal | kat | mol · s –1 | |
Units derived from SI base units may be expressed in terms of the units listed in Chapter I.
In particular, derived SI units may be expressed by the special names and symbols given in the above table; for example, the SI unit of dynamic viscosity may be expressed as m –1 · kg · s –1 or N · s · m –2 or Pa · s.]
1.3. Prefixes and their symbols used to designate certain decimal multiples and submultiples U.K.
| [F3Factor | Prefix | Symbol |
|---|---|---|
| 10 24 | yotta | Y |
| 10 21 | zetta | Z |
| 10 18 | exa | E |
| 10 15 | peta | P |
| 10 12 | tera | T |
| 10 9 | giga | G |
| 10 6 | mega | M |
| 10 3 | kilo | [X3k] |
| 10 2 | hecto | [X3h] |
| 10 1 | deca | da |
| 10 -1 | deci | d |
| 10 -2 | centi | c |
| 10 -3 | milli | m |
| 10 -6 | micro | μ |
| 10 -9 | nano | n |
| 10 -12 | pico | p |
| 10 -15 | femto | f |
| 10 -18 | atto | a |
| 10 -21 | zepto | z |
| 10 -24 | yocto | y] |
The names and symbols of the decimal multiples and submultiples of the unit of mass are formed by attaching prefixes to the word ‘ gram ’ and their symbols to the symbol ‘ g ’ .
Where a derived unit is expressed as a fraction, its decimal multiples and submultiples may be designated by attaching a prefix to units in the numerator or the denominator, or in both these parts.
Compound prefixes, that is to say prefixes formed by the juxtaposition of several of the above prefixes, may not be used.
Editorial Information
X3 Substituted by Corrigendum to Directive 1999/103/EC of the European Parliament and of the Council of 24 January 2000 amending Council Directive 80/181/EEC on the approximation of the laws of the Member States relating to units of measurement (Official Journal of the European Communities L 34 of 9 February 2000).
1.4. Special authorized names and symbols of decimal multiples and submultiples of SI units U.K.
| a The two symbols ‘ I ’ and ‘ L ’ may be used for the litre unit. (Sixteenth CGPM (1979), resolution 6). | |||
| b Unit listed in the International Bureau of Weights and Measures booklet as among the units to be permitted temporarily. | |||
| Quantity | Unit | ||
|---|---|---|---|
| Name | Symbol | Value | |
| Volume | litre | 1 or L a | 1 l = 1 dm 3 = 10 -3 m 3 |
| Mass | tonne | t | 1 t = 1 Mg = 10 3 kg |
| Pressure, stress | bar | bar b | 1 bar = 10 5 Pa |
Note: U.K.
The prefixes and their symbols listed in 1.3 may be used in conjunction with the units and symbols contained in Table 1.4. U.K.
2. UNITS WHICH ARE DEFINED ON THE BASIS OF SI UNITS BUT ARE NOT DECIMAL MULTIPLES OR SUBMULTIPLES THEREOF U.K.
| a The character (*) after a unit name or symbol indicates that it does not appear in the lists drawn up by the CGPM, CIPM o BIPM. This applies to the whole of this Annex. | |||
| b No international symbol exists. | |||
| Quantity | Unit | ||
|---|---|---|---|
| Name | Symbol | Value | |
| Plane angle | revolution* a b | 1 revolution = 2 π rad | |
| grade* or gon* | gon* | 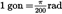 | |
| degree | ° | 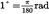 | |
| minute of angle | ′ |  | |
| second of angle | ″ | 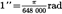 | |
| Time | minute | min | 1 min = 60 s |
| hour | h | 1 h = 3 600 s | |
| day | d | 1 d = 86 400 s | |
Note: U.K.
The prefixes listed in 1.3 may only be used in conjunction with the names ‘ grade ’ or ‘ gon ’ and the symbol ‘ gon ’ . U.K.
[F33. UNITS USED WITH THE SI, WHOSE VALUES IN SI ARE OBTAINED EXPERIMENTALLY U.K.
| Note: | |||
| The prefixes and their symbols listed in 1.3 may be used in conjunction with these two units and with their symbols.] | |||
| Quantity | Unit | ||
|---|---|---|---|
| Name | Symbol | Definition | |
| Energy | Electronvolt | eV | The electron volt is the kinetic energy acquired by an electron in passing through a potential difference of 1 volt in vaccum |
| Mass | Unified atomic mass unit | u | The unified atomic mass units is equal to 1/12 of the mass of an atom of the nuclide 12 C. |
4. UNITS AND NAMES OF UNITS PERMITTED IN SPECIALIZED FIELDS ONLY U.K.
| Quantity | Unit | ||
|---|---|---|---|
| Name | Symbol | Value | |
| Vergency of optical systems | dioptre* | 1 dioptre = 1 m -1 | |
| Mass of precious stones | metric carat | 1 metric carat = 2 × 10 -4 kg | |
| Area of farmland and building land | are | a | 1 a = 10 2 m 2 |
| Mass per unit length of textile yarns and threads | tex* | tex* | 1 tex = 10 -6 kg · m -1 |
| Blood pressure and pressure of other body fluids | Millimetre of mercury | mm Hg(*) | 1 mm Hg = 133,322 Pa |
| Effective cross-sectional area | Barn | b | 1 b = 10 -28 m 2 |
Note: U.K.
[F1The prefixes and their symbols listed in 1.3 may be used in conjunction with the above units and symbols, with the exception of the millimetre of mercury and its symbol. The multiple of 10 2 a is, however, called a ‘ hectare ’ .] U.K.
5. COMPOUND UNITS U.K.
Combinations of the units listed in Chapter I form compound units.]