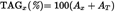- Latest available (Revised)
- Original (As adopted by EU)
Commission Implementing Regulation (EU) No 1348/2013Show full title
Commission Implementing Regulation (EU) No 1348/2013 of 16 December 2013 amending Regulation (EEC) No 2568/91 on the characteristics of olive oil and olive-residue oil and on the relevant methods of analysis
- Show Geographical Extent(e.g. England, Wales, Scotland and Northern Ireland)
- Show Timeline of Changes
More Resources
When the UK left the EU, legislation.gov.uk published EU legislation that had been published by the EU up to IP completion day (31 December 2020 11.00 p.m.). On legislation.gov.uk, these items of legislation are kept up-to-date with any amendments made by the UK since then.
This item of legislation originated from the EU
Legislation.gov.uk publishes the UK version. EUR-Lex publishes the EU version. The EU Exit Web Archive holds a snapshot of EUR-Lex’s version from IP completion day (31 December 2020 11.00 p.m.).
Changes over time for:
Changes to legislation:
This version of this Regulation was derived from EUR-Lex on IP completion day (31 December 2020 11:00 p.m.). It has not been amended by the UK since then. Find out more about legislation originating from the EU as published on legislation.gov.uk.![]()
Changes to Legislation
Revised legislation carried on this site may not be fully up to date. At the current time any known changes or effects made by subsequent legislation have been applied to the text of the legislation you are viewing by the editorial team. Please see ‘Frequently Asked Questions’ for details regarding the timescales for which new effects are identified and recorded on this site.
“ANNEX XXa METHOD FOR THE DETECTION OF EXTRANEOUS OILS IN OLIVE OILS
1.SCOPEU.K.
This method is used to detect the presence of extraneous vegetable oils in olive oils. High linoleic vegetable oils (soybean, rapeseed, sunflower, etc.), and some high oleic vegetable oils - such as hazelnut, high oleic sunflower and olive-pomace oils - can be detected in olive oils. The level detected depends on the type of extraneous oil and the variety of olive. For hazelnut oil, a detection level between 5 and 15 % is common. The method is unable to identify the type of extraneous oil detected, and only indicates if the olive oil is genuine or non-genuine.
2.PRINCIPLEU.K.
The oil is purified by solid phase extraction (SPE) on silica gel cartridges. The triacylglycerol (TAG) composition is determined by reverse phase high resolution liquid chromatography using a refractive index detector and propionitrile as the mobile phase. Fatty acid methyl esters (FAMEs) are prepared from purified oil by methylation with a cold solution of KOH in methanol (Annex X B) and then the esters are analysed by capillary gas chromatography using high polar columns (Annex X A). The theoretical triacylglycerol composition is calculated from the fatty acid composition by a computer program assuming a 1,3-random, 2-random distribution of fatty acids in the triacylglycerol, with restrictions for saturated fatty acids in the 2-position. The calculation method is a modification of the procedure described in Annex XVIII. Several mathematical algorithms are calculated from theoretical and experimental (HPLC) triacylglycerol compositions, and the resulting values are compared with those contained in a database built from genuine olive oils.
3.MATERIAL AND REAGENTSU.K.
3.1. Oil purification U.K.
3.1.1.25-ml conical flasks.U.K.
3.1.2.5-ml screw top glass tubes and caps fitted with PTFE joint.U.K.
3.1.3.Silica gel cartridges, 1 g (6 ml), for solid phase extraction (for example, Waters, Massachusetts, USA).U.K.
3.1.4. n-hexane, analytical grade.U.K.
3.1.5.Solvent mixture of hexane/diethyl ether (87:13, v/v).U.K.
3.1.6. N-heptane, analytical grade.U.K.
3.1.7.Acetone, analytical grade.U.K.
3.2. HPLC analysis of triacylglycerols U.K.
3.2.1.Micro syringes (50 μL) and needles for HPLC injection.U.K.
3.2.2.Propionitrile, super purity or HPLC grade (for example, ROMIL, Cambridge, United Kingdom), used as mobile phase.U.K.
3.2.3.HPLC column (25 cm × 4 mm internal diameter), packed with RP-18 phase (4 μm particle size).U.K.
3.3. Preparation of fatty acid methyl esters U.K.
(See Annex X B)
3.3.1.Methanol containing not more than 0,5 % water.U.K.
3.3.2.Heptane, analytical grade.U.K.
3.3.3.A 2N solution of potassium hydroxide in methanol. Dissolve 1,1 g of potassium hydroxide in 10 ml of methanol.U.K.
3.3.4.5-ml screw top glass tubes and caps provided with PTFE joint.U.K.
3.4. GC analysis of FAMEs U.K.
(See method for the determination of trans-unsaturated fatty acids by capillary column gas chromatography set out in Annex X A).
3.4.1Micro syringes (5 μL) and needles for GC injection.U.K.
3.4.2Hydrogen or helium as carrier gas.U.K.
3.4.3Hydrogen and oxygen for FID detector.U.K.
3.4.4Nitrogen or helium as auxiliary carrier gas.U.K.
3.4.5.Fused silica capillary column (50-60 m × 0,25 – 0,30 mm internal diameter) coated with cyanopropylpolysiloxane or cyanopropylphenylsiloxane phases (SP-2380 or similar) with 0,20-0,25 μm of film thickness.U.K.
4.APPARATUSU.K.
4.1.Vacuum apparatus for solid phase extraction.U.K.
4.2.Rotary evaporator.U.K.
4.3.HPLC equipment composed of:U.K.
Degasser for the mobile phase.
Rheodyne injector valve with a 10 μL loop.
High pressure pump unit.
Thermostatic oven for the HPLC column capable of maintaining sub-ambient temperatures (15-20 °C), (for example, Peltier type).
Refractive index detector.
Computerised data acquisition system provided with an integration program.
4.4Capillary gas chromatography equipment described in Annex X A, provided with:U.K.
Split injector.
Flame ionisation detector (FID).
Oven with programmable temperature.
Computerised data acquisition system provided with an integration program.
4.5.Computer with Microsoft EXCEL program.U.K.
5.ANALYTICAL PROCEDUREU.K.
5.1. Oil purification U.K.
An SPE silica gel cartridge is placed in a vacuum elution apparatus and washed under vacuum with 6 ml of hexane. The vacuum is released to prevent the column from drying and a conical flask is placed under the cartridge. A solution of the oil (0,12 g, approximately) in 0,5 ml of hexane is loaded into the column and the solution is pulled through and then eluted with 10 ml of the solvent mixture (3.1.5) of hexane-diethyl ether (87:13 v/v) under vacuum. The eluted solvent is homogenised and approximately half of the volume is poured into another conical flask. Both solutions are separately evaporated to dryness in a rotary evaporator under reduced pressure at room temperature. For triacylglycerol analysis, one of the residues is dissolved in 1 ml of acetone (See first paragraph of point 5.2) and poured into a 5-ml screw top glass tube. The other residue is dissolved in 1 ml of n-heptane and poured into a second 5-ml screw top glass tube for preparing the fatty acid methyl esters.
Note: Oil purification may be done using a silica gel column, as described in IUPAC method 2.507.U.K.
5.2. HPLC analysis of triacylglycerols U.K.
Set up the HPLC system, maintaining the column temperature at 20 °C and using propionitrile as the mobile phase at a flow rate of 0,6 ml/min. When the baseline is stable run a solvent injection; if the base line appears disturbed in the region from 12 to 25 min, use another type of acetone or a mixture of propionitrile/acetone (25:75) to dissolve the sample.
Note: Some types of acetone produce disturbances of the baseline in the above-mentioned region.U.K.
Inject a 10 μl aliquot of the solution of purified oil in acetone (5 %). The run takes approximately 60 min. Oven temperature and/or flow rate must be adjusted to achieve a chromatogram similar to that depicted in Figure 1 where trilinolein (peak 1) elutes at 15,5 min and the resolutions between the pairs LLL/OLLn (peaks 1 and 2) and OLL/OOLn (peaks 4 and 5) are good.
The height of peak 2 (OLLn+PoLL) must reach at least 3 % of the full scale.
5.3. Preparation of fatty acid methyl esters U.K.
Add 0,1 mL of a 2N solution of potassium hydroxide in methanol to the solution of purified oil in 1 mL of n-heptane. Cap the tube and screw tight. Shake the tube vigorously for 15 seconds and leave to stratify until the upper layer becomes clear (5 minutes). The n-heptane solution is ready to be injected into the gas chromatograph. The solution may be left at room temperature for a maximum of 12 hours.
5.4. GC analysis of fatty acid methyl esters U.K.
The procedure described in the method for the determination of trans-unsaturated fatty acids must be used (see Annex X A).
The GC system is set up at an oven temperature of 165 °C. The recommended oven temperature is isothermal at 165 °C for 10 min, then raising it to 200 °C at 1,5 °C/min. An injector temperature between 220 °C and 250 °C is recommended to minimise the formation of trans-fatty acids (see Annex X A). Detector temperature 250 °C. Hydrogen or helium must be used as the carrier gas at a column head pressure of 130 kPa, approximately. Injection volume 1μL in split injection mode.
A GC profile similar to that shown in Figure 2 must be obtained. Special attention must be paid to the resolution between C18:3 and C20:1 (the C18:3 peak must appear before the C20:1). To achieve these conditions, the initial temperature and/or the column head pressure must be optimised. Adjust the injector conditions (temperature, split ratio and volume injection) to minimise the discrimination of palmitic and palmitoleic acid.
The height of the C20:0 peak must be about 20 % of full scale to quantify the trans isomers. If the C18:0 peak appears distorted, reduce the sample amount.
6.INTEGRATION OF CHROMATOGRAPHIC PEAKSU.K.
6.1. HPLC chromatogram U.K.
Figure 1 shows a typical HPLC chromatogram of the triacylglycerols of a purified olive oil. For peak integration, three baselines must be traced: the first between the start of peak 1 and the end of peak 3; the second between the start of peak 4 and the valley before peak 8; the third between the valley preceding peak 8 and the end of peak 18.
The total area is the sum of the areas of all the peaks (identified and not identified) from peak 1 to peak 18. The percentage of each peak is given by
The percentages have to be given to two decimal figures.
6.2. GC chromatogram U.K.
Figure 2 shows a GC chromatogram of fatty acid alkyl esters obtained from a purified olive oil. Percentages of the following fatty acids must be calculated:
| Palmitic; | P (C16:0) | = | methyl ester + ethyl ester |
| Stearic; | S (C18:0) | = | methyl ester |
| Palmitoleic; | Po (C16:1) | = | sum of methyl esters of the two cis-isomers |
| Oleic; | O (C18:1) | = | sum of methyl esters of the two cis-isomers + ethyl ester + trans-isomers |
| Linoleic; | L (C18:2) | = | methyl ester+ ethyl ester + trans-isomers |
| Linolenic; | Ln (C18:3) | = | methyl ester + trans-isomers |
| Arachidic; | A (C20:0) | = | methyl ester |
| Eicosenoic (gondoic); | G (C20:1) | = | methyl ester |
Ethyl and trans-isomers esters may be absent in the GC chromatogram.
Total area (AT) is the sum of all the peaks appearing in the chromatogram from C14:0 to C24:0, except that corresponding to squalene. The percentage of each peak is calculated as follows:
The results have to be expressed to two decimal places.
For the calculations of the computer programs, it is not necessary to normalise to 100 because this is done automatically.
Figure 1
HPLC chromatogram of TAGs of a “Chamlali” virgin olive oil. Main components of chromatographic peaks

Table 1
Repeatability data of the determination of virgin olive oil TAGs by HPLC at a column temperature of 20 °C and using propionitrile as mobile phase
| n = 3 replicates RSDr = Relative Standard Deviation of the repeatability | ||||||||||||
| ECN | HPLC peaks | TAGs | Sample 1 | Sample 2 | Sample 3 | Sample 4 | Sample 5 | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean (%) | RSDr (%) | Mean (%) | RSDr (%) | Mean (%) | RSDr (%) | Mean (%) | RSDr (%) | Mean (%) | RSDr (%) | |||
| 42 | 1 | LLL | 0,02 | 7,23 | 0,066 | 5,18 | 0,095 | 4,1 | 0,113 | 0,95 | 0,34 | 1,05 |
| 2 | OLLn+ PoLL | 0,085 | 7,44 | 0,24 | 1,78 | 0,26 | 2,25 | 0,35 | 2,02 | 0,5 | 2,83 | |
| 3 | PLLn | 0,023 | 15,74 | 0,039 | 5,51 | 0,057 | 5,62 | 0,082 | 4,35 | 0,12 | 6,15 | |
| 44 | 4 | OLL | 0,47 | 1,52 | 1,53 | 0,42 | 2,62 | 0,98 | 3,35 | 1,05 | 4,37 | 1,13 |
| 5 | OOLn+ PoOL | 1,07 | 2,01 | 1,54 | 0,46 | 1,61 | 0,71 | 1,72 | 1,07 | 1,77 | 2,4 | |
| 6 | PLL+ PoPoO | 0,11 | 12,86 | 0,24 | 4,37 | 0,65 | 1,32 | 1,35 | 0,73 | 2,28 | 1,24 | |
| 7 | POLn+ PpoPo+ PpoL | 0,42 | 5,11 | 0,49 | 2,89 | 0,55 | 2,01 | 0,85 | 1,83 | 1,09 | 1,96 | |
| 46 | 8 | OOL+ LnPP | 6,72 | 0,63 | 8,79 | 0,31 | 11,21 | 0,42 | 13,25 | 0,33 | 15,24 | 0,23 |
| 9 | PoOO | 1,24 | 2,86 | 1,49 | 0,95 | 1,63 | 0,85 | 2,12 | 0,45 | 2,52 | 0,56 | |
| 10 | SLL+ PLO | 2,7 | 0,65 | 4,05 | 0,7 | 6,02 | 0,65 | 9,86 | 0,53 | 11,53 | 0,31 | |
| 11 | PoOP+ SpoL+ SOLn+ SpoPo | 0,64 | 4,42 | 0,69 | 3,02 | 0,79 | 1,23 | 1,53 | 0,89 | 1,7 | 1,66 | |
| 48 | 12+13 | OOO+ PLP+ PoPP | 49,6 | 0,07 | 48,15 | 0,06 | 42,93 | 0,06 | 33,25 | 0,1 | 24,16 | 0,06 |
| 14 | SOL | 0,82 | 1,72 | 0,92 | 1,56 | 1,05 | 1,32 | 1,25 | 1,05 | 1,6 | 1,77 | |
| 15 | POO | 22,75 | 0,25 | 21,8 | 0,2 | 21,05 | 0,3 | 20,36 | 0,35 | 20,17 | 0,14 | |
| 50 | 16 | POP | 3,05 | 0,46 | 4,56 | 0,42 | 4,98 | 0,52 | 5,26 | 0,41 | 5,57 | 0,38 |
| 17 | SOO | 6,87 | 0,21 | 5,56 | 0,33 | 4,86 | 0,43 | 4,12 | 0,72 | 3,09 | 0,69 | |
| 18 | POS+ SLS | 1,73 | 1,23 | 1,65 | 1,1 | 1,54 | 0,99 | 1,49 | 1,1 | 1,41 | 1,0 | |
Figure 2
GC chromatogram of fatty acid alkyl esters obtained from an olive-pomace oil by transesterification with a cold solution of KOH in methanol

7.DETECTION OF EXTRANEOUS OILS IN OLIVE OILSU.K.
The calculation method of the detection of extraneous oils in olive oils by means of a comparison of mathematical algorithms with a data base built from genuine olive oils is set out in Annex I to standard IOC/T.20/Doc. No 25.”
Options/Help
Print Options
PrintThe Whole Regulation
PrintThe Whole Annex
You have chosen to open the Whole Regulation
The Whole Regulation you have selected contains over 200 provisions and might take some time to download. You may also experience some issues with your browser, such as an alert box that a script is taking a long time to run.
Would you like to continue?
You have chosen to open Schedules only
The Schedules you have selected contains over 200 provisions and might take some time to download. You may also experience some issues with your browser, such as an alert box that a script is taking a long time to run.
Would you like to continue?
Legislation is available in different versions:
Latest Available (revised):The latest available updated version of the legislation incorporating changes made by subsequent legislation and applied by our editorial team. Changes we have not yet applied to the text, can be found in the ‘Changes to Legislation’ area.
Original (As adopted by EU): The original version of the legislation as it stood when it was first adopted in the EU. No changes have been applied to the text.
See additional information alongside the content
Geographical Extent: Indicates the geographical area that this provision applies to. For further information see ‘Frequently Asked Questions’.
Show Timeline of Changes: See how this legislation has or could change over time. Turning this feature on will show extra navigation options to go to these specific points in time. Return to the latest available version by using the controls above in the What Version box.
More Resources
Access essential accompanying documents and information for this legislation item from this tab. Dependent on the legislation item being viewed this may include:
- the original print PDF of the as adopted version that was used for the EU Official Journal
- lists of changes made by and/or affecting this legislation item
- all formats of all associated documents
- correction slips
- links to related legislation and further information resources
Timeline of Changes
This timeline shows the different versions taken from EUR-Lex before exit day and during the implementation period as well as any subsequent versions created after the implementation period as a result of changes made by UK legislation.
The dates for the EU versions are taken from the document dates on EUR-Lex and may not always coincide with when the changes came into force for the document.
For any versions created after the implementation period as a result of changes made by UK legislation the date will coincide with the earliest date on which the change (e.g an insertion, a repeal or a substitution) that was applied came into force. For further information see our guide to revised legislation on Understanding Legislation.
More Resources
Use this menu to access essential accompanying documents and information for this legislation item. Dependent on the legislation item being viewed this may include:
- the original print PDF of the as adopted version that was used for the print copy
- correction slips
Click 'View More' or select 'More Resources' tab for additional information including:
- lists of changes made by and/or affecting this legislation item
- confers power and blanket amendment details
- all formats of all associated documents
- links to related legislation and further information resources