- Latest available (Revised)
- Original (As made)
The Food Additives and Novel Foods (Authorisations and Miscellaneous Amendments) and Food Flavourings (Removal of Authorisations) (Scotland) Regulations 2024
You are here:
- Scottish Statutory Instruments
- 2024 No. 156
- Schedules only
More Resources
Status:
This is the original version (as it was originally made). This item of legislation is currently only available in its original format.
Regulation 3
SCHEDULE 1Amendments to Annex 2 (domestic list of food additives approved for use in foods and conditions for use) to Regulation (EC) No 1333/2008 concerning steviol glycosides from Stevia (E 960a – E 960c) and the extension of use of polyglycerol polyricinoleate (E 476)
1. In Part B (list of all additives), in paragraph 2 (sweeteners) after the entry for E 960a (Steviol glycosides from Stevia), insert—
| “E 960b | Steviol glycosides from fermentation”. |
2. In Part C (definitions of groups of additives), in sub-part 5 (other additives that may be regulated combined), in paragraph (v)—
(a)for the heading of the paragraph, substitute “E 960a – E 960c: Steviol glycosides”,
(b)after the entry for E 960a (Steviol glycosides from Stevia), insert—
| “E 960b | Steviol glycosides from fermentation”. |
3. In Part E (authorised food additives and conditions of use in food categories), in the table—
(a)in each place it occurs, for “E 960a and E 960c” substitute “E 960a – E 960c”,
(b)in category 03 (edible ices), after the entry for E 473-474 (sucrose esters of fatty acids – sucroglycerides), insert—
| “E 476 | Polyglycerol polyricinoleate | 4000 | except sorbets”, |
(c)in category 05.1 (cocoa and chocolate products), at the end, insert the following footnote—
“(1) The additives may be added individually or in combination.”,
(d)in category 05.2 (other confectionary including breath freshening microsweets)—
(i)in the third entry for Group IV (polyols), for “only cocoa or dried fruit-based, milk or fat-based sandwich spreads, energy-reduced or with no added sugar” substitute “sandwich spreads made with a base of cocoa, milk, dried fruit or fat; energy reduced or with no added sugar”,
(ii)in the first entry for E 960a – E960c Steviol glycosides(1) for “only cocoa or dried-fruit-based, energy-reduced or with no added sugar”, substitute “only cocoa or dried fruit based; energy reduced or with no added sugar”,
(iii)in the second entry for E 960a – E960c Steviol glycosides for “only cocoa, milk, dried-fruit-based or fat-based sandwich spreads, energy-reduced or with no added sugar”, substitute “sandwich spreads made with a base of cocoa, milk, dried fruit or fat; energy reduced or with no added sugar”,
(e)in category 05.4 (decorations, coatings and fillings, except fruit-based fillings covered by category 4.2.4), for the second entry for E 960a – E 960c Steviol glycosides for “only cocoa or dried-fruit-based, energy-reduced or with no added sugar”, substitute “only cocoa or dried fruit based; energy reduced or with no added sugar”,
(f)in category 12.6 (sauces), for the entry for “E 476” (polyglycerol polyricinoleate), substitute—
| “E 476 | Polyglycerol polyricinoleate | 4000 | only emulsified sauces with a fat content of less than 20% | |
| E 476 | Polyglycerol polyricinoleate | 8000 | only emulsified sauces with a fat content of 20% or more”. |
Regulation 4(4)
SCHEDULE 2Amendment to the Annex to Commission Regulation (EU) No 231/2012 for the authorisation of steviol glycosides from fermentation (Yarrowia lipolytica) (E 960b)
1. In the appropriate place, insert the following entry—
“E 960b STEVIOL GLYCOSIDES FROM FERMENTATION (YARROWIA LIPOLYTICA)
| Synonyms | |||
|---|---|---|---|
| Definition | Steviol glycosides from Yarrowia lipolytica consist of a mixture predominantly composed of rebaudioside M, with some rebaudioside D, and smaller amounts of rebaudioside A and rebaudioside B. The manufacturing process comprises two main phases. The first phase involves fermentation of a non-toxigenic non-pathogenic strain of Yarrowia lipolytica VRM that has been genetically modified with heterologous genes to overexpress steviol glycosides. Removal of biomass by solid-liquid separation and heat treatment is followed by concentration of the steviol glycosides. The second phase involves purification by employing ion-exchange chromatography, followed by recrystallisation of the steviol glycosides resulting in a final product containing not less than 95% of rebaudiosides M, D, A, and B. Viable cells or the DNA of Yarrowia lipolytica VRM must not be detected in the food additive. | ||
| Chemical name | Rebaudioside A: 13-[(2-O-β-D-glucopyranosyl-3-O-β-D-glucopyranosyl-β-D-glucopyranosyl)oxy]kaur-16-en-18-oic acid, β-D-glucopyranosyl ester Rebaudioside B: 13-[(2-O-β–D-glucopyranosyl-3-O-β– D-glucopyranosyl-β-D-glucopyranosyl)oxy]kaur-16-en-18-oic acid Rebaudioside D: 13-[(2-O-β-D-glucopyranosyl-3-O-β-D-glucopyranosyl-β-D-glucopyranosyl)oxy]kaur-16-en-18-oic acid, 2-O-β-D-glucopyranosyl-β-D-glucopyranosyl ester Rebaudioside M: 13-[(2-O-β-D-glucopyranosyl-3-O-β-D-glucopyranosyl-β-D-glucopyranosyl)oxy]kaur-16-en-18-oic acid, 2-O-β-D-glucopyranosyl-3-O-β-D-glucopyranosyl-β-D-glucopyranosyl ester | ||
| Molecular formula | Trivial name | Formula | Conversion factor |
| Rebaudioside A | C44 H70 O23 | 0.33 | |
| Rebaudioside B | C38 H60 O18 | 0.40 | |
| Rebaudioside D | C50 H80 O28 | 0.29 | |
| Rebaudioside M | C56 H90 O33 | 0.25 | |
| Molecular weight and CAS No | Trivial name | CAS Number | Molecular weight (g/mol) |
| Rebaudioside A | 58543-16-1 | 967.01 | |
| Rebaudioside B | 58543-17-2 | 804.88 | |
| Rebaudioside D | 63279-13-0 | 1129.15 | |
| Rebaudioside M | 1220616-44-3 | 1291.29 | |
| Assay | Not less than 95 % of rebaudioside A, rebaudioside B, rebaudioside D and rebaudioside M on the dried basis | ||
| Description | White to light yellow powder, approximately between 200 and 350 times sweeter than sucrose (at 5 % sucrose equivalency) | ||
| Identification | |||
| Solubility | Freely soluble to slightly soluble in water | ||
| pH | Between 4.5 and 7.0 (1 in 100 solution) | ||
| hPurity | |||
| Total ash | Not more than 1 % | ||
| Loss on drying | Not more than 6 % (105°C, 2h) | ||
| Residual solvent | Not more than 5000 mg/kg ethanol | ||
| Arsenic | Not more than 0.1 mg/kg | ||
| Lead | Not more than 0.1 mg/kg | ||
| Cadmium | Not more than 0.01 mg/kg | ||
| Mercury | Not more than 0.05 mg/kg | ||
| Residual protein | Not more than 20 mg/kg | ||
| Microbiological criteria | |||
| Total (aerobic) plate count | Not more than 1000 CFU/g | ||
| Yeast | Not more than 100 CFU/g | ||
| Moulds | Not more than 100 CFU/g | ||
| Escherichia coli | Negative in 1g | ||
| Salmonella spp. | Negative in 25g”. | ||
Regulation 4(6)
SCHEDULE 3Amendment to the Annex to Commission Regulation (EU) No 231/2012 for the re-numbering of rebaudioside M produced via enzyme modification of steviol glycosides from Stevia E 960c(i) (formerly E 960c) and for the addition of a specification for rebaudioside M, AM and D produced via enzymatic conversion of highly purified steviol glycosides from Stevia leaf extracts (E 960c(ii))
1. In the appropriate place, insert the following entry—
“E 960C(ii) REBAUDIOSIDE M, AM AND D PRODUCED VIA ENZYMATIC CONVERSION OF HIGHLY PURIFIED STEVIOL GLYCOSIDES FROM STEVIA LEAF EXTRACTS
| Synonyms | |||
|---|---|---|---|
| Definition | Steviol glycosides produced via enzymatic conversion of highly purified steviol glycosides (rebaudioside A or stevioside) from Stevia leaf extracts are composed predominantly of rebaudioside M, rebaudioside D, and rebaudioside AM. Rebaudiosides D, M and AM are produced via enzymatic conversion of highly purified steviol glycoside (rebaudioside A or stevioside) extracts (95% steviol glycosides) obtained from Stevia rebaudiana Bertoni plant using UDP-glucosyltransferase and sucrose synthase enzymes produced by genetically modified strains of Escherichia coli (pPM294, pFAH170, and pSK041) that facilitate the transfer of glucose from sucrose and UDP-glucose to steviol glycosides via glycosidic bonds. After removal of the enzymes by solid-liquid separation and heat treatment, the purification involves concentration of the steviol glycosides by resin adsorption, followed by recrystallisation of the steviol glycosides resulting in a final product containing not less than 95 % of total steviol glycosides, including one or more of rebaudiosides D, M and AM. Viable cells or DNA of Escherichia coli (pPM294, pFAH170, and pSK041) must not be detected in the food additive. | ||
| Chemical name | Rebaudioside M: 13-[(2-O-β-D-glucopyranosyl-3-O-β-D-glucopyranosyl-β-D-glucopyranosyl)oxy]kaur-16-en-18-oic acid, 2-O-β-D-glucopyranosyl-3-O-β-D-glucopyranosyl-β-D-glucopyranosyl ester Rebaudioside D: 13-[(2-O-β-D-glucopyranosyl-3-O-β-D-glucopyranosyl-β-D-glucopyranosyl)oxy]kaur-16-en-18-oic acid, 2-O-β-D-glucopyranosyl-β-D-glucopyranosyl ester Rebaudioside AM: 13-[(2-O-β-D-glucopyranosyl-β-D-glucopyranosyl)oxy]kaur-16-en-18-oic acid, 2-O-β-D-glucopyranosyl-3-O-β-D-glucopyranosyl-β-D-glucopyranosyl ester | ||
| Molecular formula | Trivial name | Formula | Conversion factor |
| Rebaudioside M | C56 H90 O33 | 0.25 | |
| Rebaudioside D | C50 H80 O28 | 0.29 | |
| Rebaudioside AM | C50 H80 O28 | 0.29 | |
| Molecular weight and CAS No | Trivial name | CAS Number | Molecular weight (g/mol) |
| Rebaudioside M | 1220616-44-3 | 1291.29 | |
| Rebaudioside D | 63279-13-0 | 1129.15 | |
| Rebaudioside AM | 2222580-26-7 | 1129.15 | |
| Assay | Not less than 95 % of steviol glycosides on the dried basis, including one or more of rebaudiosides D, M and AM | ||
| Description | White to light yellow powder, approximately between 200 and 350 times sweeter than sucrose (at 5 % sucrose equivalency) | ||
| Identification | |||
| Solubility | Freely soluble to slightly soluble in water | ||
| pH | Between 4.5 and 7.0 (1 in 100 solution) | ||
| Purity | |||
| Total ash | Not more than 1 % | ||
| Loss on drying | Not more than 6 % (105°C, 2h) | ||
| Residual solvent | Not more than 5000 mg/kg ethanol | ||
| Arsenic | Not more than 0.015 mg/kg | ||
| Lead | Not more than 0.2 mg/kg | ||
| Cadmium | Not more than 0.015 mg/kg | ||
| Mercury | Not more than 0.07 mg/kg | ||
| Residual protein | Not more than 5 mg/kg”. | ||
Regulation 5
SCHEDULE 4Amendments to the Annex (domestic list of novel foods authorised to be placed on the market within Great Britain) to Commission Implementing Regulation (EU) 2017/2470 for the authorisation of partially hydrolysed protein from spent barley (Hordeum vulgare) and rice (Oryza sativa) as a novel food
1. In Table 1 (authorised novel foods), after the entry for Astaxanthin-rich oleoresin from Haematococcus pluvialis algae insert the following entry—
2. In Table 2 (specifications), after the entry for Astaxanthin-rich oleoresin from Haematococcus pluvialis algae insert the following entry—
| “Partially hydrolysed protein from spent barley (Hordeum vulgare) and rice (Oryza sativa) | Description/Definition: |
|---|---|
| Partially hydrolysed protein from spent barley (Hordeum vulgare) and rice (Oryza sativa) is an off-white powder, produced by concentration of proteins from a mixture of barley and rice from the mash step of beer production using a series of enzymatic hydrolysis and mechanical purification steps. Characteristics/Composition: Protein (dry basis): ≥ 85% Moisture: <8% Total Carbohydrates: <10% Fat: <2% Ash: <8% Heavy metals: Arsenic: ≤0.1 mg/kg Cadmium: <0.1 mg/kg Lead: <0.2 mg/kg Mercury: <0.1 mg/kg Microbiological criteria: Aerobic plate count: <30,000 CFU/g Coliforms: <10 CFU/g Yeast and Mould: <50 CFU/g Salmonella spp: Negative in 25 g Escherichia coli: <10 CFU/g Staphylococcus aureus: <10 CFU/g Listeria spp.: Negative in 25 g CFU: Colony Forming Units”. |
Regulation 5
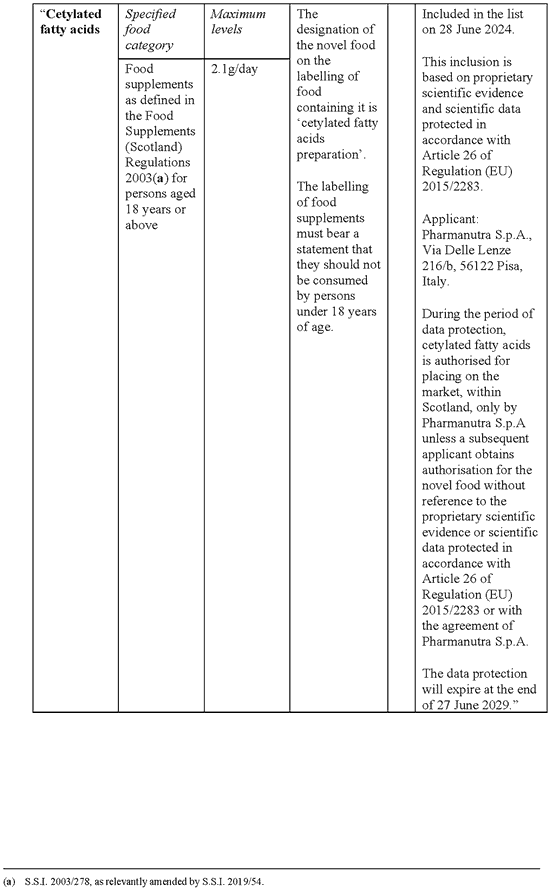
SCHEDULE 5Amendments to the Annex (domestic list of novel foods authorised to be placed on the market within Great Britain) to Commission Implementing Regulation (EU) 2017/2470 for the authorisation of cetylated fatty acids as a novel food
1. In Table 1 (authorised novel foods), after the entry for Calanus finmarchicus oil insert the following entry—
2. In Table 2 (specifications), after the entry for Calanus finmarchicus oil insert the following entry—
| “Cetylated fatty acids | Description/Definition: The novel food is a mixture of 70 – 80% cetylated fatty acids which are produced from the reaction of cetyl alcohol with myristic acid and oleic acid. Characteristics/Composition: Physical status at 25°C: Solid Colour (APHA Colour): ≤ 600 Acid value (mg KOH/g): ≤ 5 Iodine value (I2g/100g): 30 – 50 Saponification value (mg KOH/g): 130 – 150 Hydroxyl value (mg KOH/g): ≤ 20 Ester content (%): 70 – 80 Cetyl oleate (%): 22 – 30 Cetyl myristate (%): 41 – 56 Triglycerides (%): 22 – 25 Microbiological criteria: Total aerobic microbial count (CFU/g): ≤ 1000 Yeasts and moulds (CFU/g): ≤ 100 APHA: American Public Health Association CFU: Colony Forming Units KOH: potassium hydroxide”. |
|---|
Regulation 5
SCHEDULE 6Amendments to the Annex (domestic list of novel foods authorised to be placed on the market within Great Britain) to Commission Implementing Regulation (EU) 2017/2470 for the authorisation of 3-fucosyllactose (3-FL) (produced by a derivative strain of Escherichia coli K-12 DH1) as a novel food
1. In Table 1 (authorised novel foods), after the entry for 2’-Fucosyllactose / Difucosyllactose mixture (‘2’-FL/DFL’) (microbial source) insert the following entry—
2. In Table 2 (specifications), after the entry for 2’-Fucosyllactose / Difucosyllactose mixture (‘2’-FL/DFL’) (microbial source) insert the following entry—
| “3-Fucosyllactose (3-FL) (produced by a derivative strain of Escherichia coli K-12 DH1) | Description/Definition: |
|---|---|
| 3-Fucosyllactose (3-FL) (produced by a derivative strain of Escherichia coli K-12 DH1) is a purified carbohydrate powder or agglomerate containing at least 90% of 3-fucosyllactose on a dry matter basis obtained from microbial fermentation with a genetically modified strain of Escherichia coli K-12 DH1. Chemical name: β-D-Galactopyranosyl-(1→4)- [α-L-fucopyranosyl-(1→3)]- D-glucopyranose Chemical formula: C18H32O15 Molecular mass: 488.44 Da CAS No: 41312-47-4 Characteristics/Composition: Appearance: Powder, agglomerates, powder with agglomerates Colour: White to off-white Assay (water free) – Specified saccharides (includes 3-FL, D-lactose, L-fucose and 3-fucosyllactose): ≥ 92.0 w/w % Assay (water free) – 3-FL: ≥ 90.0 w/w % L-Fucose: ≤ 1.0 w/w % D-Lactose: ≤ 5.0 w/w % 3-Fucosyllactulose: ≤ 1.5 w/w % Sum of other carbohydrates: ≤ 5.0 w/w % pH in 5% solution (20°C): 3.2–7.0 Water: ≤ 6.0 w/w% Ash, sulphated: ≤ 0.5 w/w % Acetic acid (relevant only for crystallised 3-FL) : ≤ 1.0 w/w % Residual protein by Bradford assay: ≤ 0.01 w/w % Residual endotoxins: ≤ 10 EU/mg Heavy metals: Lead: ≤ 0.1 mg/kg Arsenic: ≤ 0.2 mg/kg Mycotoxins: Aflatoxin M1: ≤ 0.025 µg/kg Microbiological criteria: Aerobic mesophilic total plate count: ≤ 1000 CFU/g Enterobacteriaceae: absent in 10g Salmonella spp: absent in 25g Bacillus cereus: ≤ 50 CFU/g Listeria monocytogenes: absent in 25g Cronobacter spp.: absent in 10g Yeasts: ≤ 100 CFU/g Moulds: ≤ 100 CFU/g EU: Endotoxin Units CFU: Colony Forming Units”. |
Regulation 5
SCHEDULE 7Amendments to the Annex (domestic list of novel foods authorised to be placed on the market within Great Britain) to Commission Implementing Regulation (EU) 2017/2470 for the authorisation of lacto-N-fucopentaose I (LNFP-I) and 2’-fucosyllactose (2’-FL) mixture as a novel food
1. In Table 1 (authorised novel foods), after the entry for Lactitol insert the following entry—
2. In Table 2 (specifications), after the entry for Lactitol insert the following entry—
| “Lacto-N-fucopentaose I (LNFP-I) and 2’-fucosyllactose (2’-FL) mixture | Description/Definition: |
|---|---|
| Lacto-N-fucopentaose I (LNFP-I) and 2’-fucosyllactose (2’-FL) mixture is a purified carbohydrate powder or agglomerate obtained from microbial fermentation with a genetically modified strain of Escherichia coli K-12 DH1 containing at least 75% of LNFP-I and 2’-FL of dry matter, where ≥ 50% is LNFP-I (dry weight) and ≥ 15% is 2’-FL (dry weight). Characteristics/Composition: Appearance: Powder, agglomerates, powder with agglomerates Colour: White to off-white Assay (water-free) – Specified saccharides (includes LNFP-I, 2’-FL, lacto-N-tetraose, difucosyl-D-lactose, 3-fucosyllactose, D-lactose, L-fucose and 2’-fucosyl-lactitol, LNFP-I fructose isomer and 2’-fucosyl-D-lactulose): ≥ 90.0 w/w % Assay (water-free) – LNFP-I and 2’-FL: ≥ 75.0 w/w % Assay (water-free) – LNFP-I: ≥ 50.0 w/w % Assay (water-free) – 2’-FL: ≥ 15.0 w/w % Lacto-N-tetraose: ≤ 5.0 w/w% 3-Fucosyllactose: ≤ 1.0 w/w % Sum of L-fucose and 2’-fucosyl-lactitol: ≤ 1.0 w/w % D-Lactose: ≤ 10.0 w/w % Difucosyl-D-lactose: ≤ 2.0 w/w % LNFP-I fructose isomer: ≤ 1.5 w/w % 2’-Fucosyl-D-lactulose: ≤ 1.0 w/w % Sum of other carbohydrates: ≤ 6.0 w/w % pH in 5% solution (20°C): 4.0 – 7.0 Water: ≤ 8.0 w/w % Ash, sulphated: ≤ 0.5 w/w % Residual protein by Bradford assay: ≤ 0.01 w/w % Heavy metals: Arsenic: ≤ 0.2 mg/kg Mycotoxins: Residual endotoxins: ≤10 EU/mg Aflatoxin M1: ≤0.025 µg/kg Microbiological criteria: Aerobic mesophilic total plate count: ≤ 1000 CFU/g Enterobacteriaceae: Absent in 10g Salmonella spp: Absent in 25g Yeasts: ≤ 100 CFU/g Moulds: ≤ 100 CFU/g Bacillus cereus: ≤ 50 CFU/g Listeria monocytogenes: Absent in 25g Cronobacter spp.: Absent in 10g CFU: Colony Forming Units EU: Endotoxin Units”. |
Regulation 5
SCHEDULE 8Corrections to existing entries in the Annex (domestic list of novel foods authorised to be placed on the market within Great Britain) to Commission Implementing Regulation (EU) 2017/2470 concerning the authorisation of bovine milk basic whey protein isolate and xylo-oligosaccharides
1. In Table 1 (authorised novel foods), for the entry for bovine milk basic whey protein isolate substitute the following entry—
2. In Table 2 (specifications), for the entry for Xylo-oligosaccharides, in column 2 (description/definition), after the row specifying the moisture (%) content, insert—
| “Dry Material (%) | - | - | 70 -75” |
The entries for E 960a to E 960c were re-named by paragraph 3(a) of this schedule.
Options/Help
Print Options
PrintThe Whole Instrument
PrintThe Schedules only
Legislation is available in different versions:
Latest Available (revised):The latest available updated version of the legislation incorporating changes made by subsequent legislation and applied by our editorial team. Changes we have not yet applied to the text, can be found in the ‘Changes to Legislation’ area.
Original (As Enacted or Made): The original version of the legislation as it stood when it was enacted or made. No changes have been applied to the text.
Policy Note
Policy Note sets out a brief statement of the purpose of a Scottish Statutory Instrument and provides information about its policy objective and policy implications. They aim to make the Scottish Statutory Instrument accessible to readers who are not legally qualified and accompany any Scottish Statutory Instrument or Draft Scottish Statutory Instrument laid before the Scottish Parliament from July 2012 onwards. Prior to this date these type of notes existed as ‘Executive Notes’ and accompanied Scottish Statutory Instruments from July 2005 until July 2012.
More Resources
Access essential accompanying documents and information for this legislation item from this tab. Dependent on the legislation item being viewed this may include:
- the original print PDF of the as enacted version that was used for the print copy
- lists of changes made by and/or affecting this legislation item
- confers power and blanket amendment details
- all formats of all associated documents
- correction slips
- links to related legislation and further information resources
More Resources
Use this menu to access essential accompanying documents and information for this legislation item. Dependent on the legislation item being viewed this may include:
- the original print PDF of the as made version that was used for the print copy
- correction slips
Click 'View More' or select 'More Resources' tab for additional information including:
- lists of changes made by and/or affecting this legislation item
- confers power and blanket amendment details
- all formats of all associated documents
- links to related legislation and further information resources