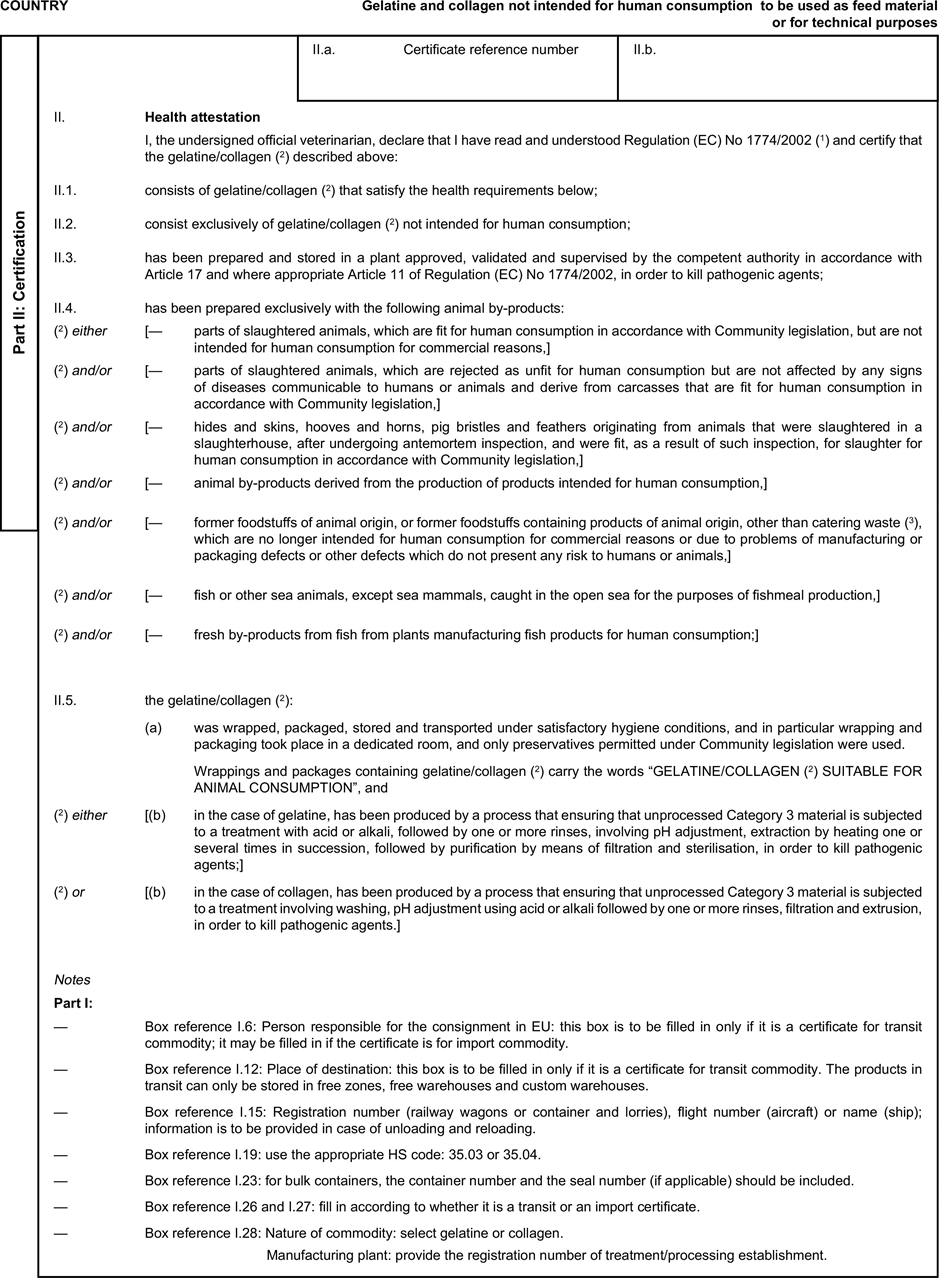
ANNEX IU.K.SPECIFIC DEFINITIONS
For the purpose of this Regulation:
1.
[‘ apiculture by-products ’ means honey, beeswax, royal jelly, propolis or pollen not intended for human consumption;]
2.
‘batch’ means a unit of production produced in a single plant using uniform production parameters — or a number of such units, when stored together — and that can be identified for the purposes of recall and re-treatment or disposal should tests show that to be necessary;
3.
‘biogas plant’ means a plant in which biological degradation of products of animal origin is undertaken under anaerobic conditions for the production and collection of biogas;
4.
‘blood products’ means products derived from blood or fractions of blood, excluding blood meal; they include dried/frozen/liquid plasma, dried whole blood, dried/frozen/liquid red cells or fractions thereof and mixtures;
5.
‘blood’ means fresh whole blood;
6.
[‘ blood meal ’ means products derived from the heat treatment of blood or fractions of blood in accordance with Annex VII, Chapter II, and intended for animal consumption or organic fertilisers;]
7.
‘canned petfood’ means heat-processed petfood contained within a hermetically sealed container;
8.
‘Category 1 or Category 2 intermediate plant’ means a plant in which unprocessed Category 1 or Category 2 material is handled and/or temporarily stored for the purpose of further transportation to its final destination and where certain preliminary activities, such as removal of hides and skins and performing post-mortem examinations, may take place;
9.
‘Category 1 processing plant’ means a plant in which Category 1 material is processed before its final disposal;
10.
‘Category 2 oleochemical plant’ means a plant processing rendered fats derived from Category 2 material under conditions set out in Annex VI, Chapter III;
11.
‘Category 2 processing plant’ means a plant in which Category 2 material is processed before its final disposal, further transformation or use;
12.
‘Category 3 intermediate plant’ means a plant in which unprocessed Category 3 material is sorted and/or cut and/or chilled or deep-frozen into blocks and/or temporarily stored for the purpose of further transporting to its final destination;
13.
‘Category 3 oleochemical plant’ means a plant processing rendered fats derived from Category 3 material;
14.
‘Category 3 processing plant’ means a plant in which Category 3 material is processed into processed animal protein and other processed products that could be used as feed material;
15.
[‘ catering waste ’ means all waste food including used cooking oil originating in restaurants, catering facilities and kitchens, including central kitchens and household kitchens;]
16.
‘co-incineration plant’ means a disposal site as defined in Article 3(5) of Directive 2000/76/EC;
17.
‘co-incineration’ means the disposal of animal by-products or products derived therefrom in a co-incineration plant;
18.
‘collection centres’ means premises collecting and treating certain animal by-products intended to be used for the feeding of the animals specified in Article 23(2)(c);
19.
‘composting plant’ means a plant in which biological degradation of products of animal origin is undertaken under aerobic conditions;
20.
‘digestion residues’ means residues resulting from the transformation of animal by-products in a biogas plant;
21.
‘digestive tract content’ means the content of the digestive tract of mammals and ratites, whether or not separated from the digestive tract;
22.
‘dogchews’ means untanned products for pet animals to chew, produced from hides and skins of ungulates or other animal material;
23.
‘feed material’ means those feed materials, as defined in Directive 96/25/EC(), that are of animal origin including processed animal proteins, blood products, rendered fats, fish oil, fat derivatives, gelatin and hydrolysed proteins, dicalcium phosphate, milk, milk-based products and colostrum;
24.
‘fishmeal’ means processed animal protein derived from sea animals, except sea mammals;
25.
‘fur animals’ means animals kept or reared for the production of fur and not used for human consumption;
26.
‘gelatin’ means natural, soluble protein, gelling or non-gelling, obtained by the partial hydrolysis of collagen produced from bones, hides and skins, tendons and sinews of animals (including fish and poultry);
27.
‘greaves’ means the protein-containing residue of rendering, after partial separation of fat and water;
28.
‘hermetically sealed container’ means a container that is designed and intended to be secure against the entry of micro-organisms;
29.
‘hides and skins’ means all cutaneous and subcutaneous tissues;
30.
‘high-capacity incineration plant’ means an incineration plant other than a low-capacity incineration plant;
31.
‘hydrolysed proteins’ means polypeptides, peptides and aminoacids, and mixtures thereof, obtained by the hydrolysis of animal by-products;
32.
‘incineration plant’ means a disposal site as defined in Article 3(4) of Directive 2000/76/EC;
33.
‘incineration’ means the disposal of animal by-products or products derived therefrom in an incineration plant;
34.
‘laboratory reagent’ means a packaged product, ready for use by the end user, containing a blood product, and intended for laboratory use as reagent or reagent product, whether used alone or in combination;
35.
‘landfill’ means a disposal site as defined by Directive 1999/31/EC;
36.
‘low-capacity incineration plant’ means an incineration plant with a throughput of less than 50 kg of animal by-products per hour;
37.
[‘ manure ’ means any excrement and/or urine of farmed animals, with or without litter, or guano, that may be either unprocessed or processed in accordance with Chapter III of Annex VIII or otherwise transformed in biogas or composting plants;]
38.
‘organic fertilizers’ and ‘soil improvers’ mean materials of animal origin used to maintain or improve plant nutrition and the physical and chemical properties and biological activity of soils, either separately or together; they may include manure, digestive tract content, compost and digestion residues;
39.
‘[pasture land’means land covered with grass or other herbage grazed by or used as feedingstuffs for farmed animals, excluding land to which organic fertilisers and soil improvers have been applied in accordance with Commission Regulation (EC) No 181/2006 () ;]
40.
‘[petfood plant’ means a plant producing petfood or dogchews or flavouring innards and in which certain animal by-products are used in the preparation of such petfood, dogchews or flavouring innards;]
41.
‘petfood’ means food for pet animals containing Category 3 material;
42.
[‘ processed animal protein ’ means animal protein derived entirely from Category 3 material, which have been treated in accordance with Chapter II of Annex VII so as to render them suitable for direct use as feed material or for any other use in feedingstuffs, including petfood, or for use in organic fertilisers or soil improvers; however, it does not include blood products, milk, milk-based products, colostrum, gelatine, hydrolysed proteins and dicalcium phosphate, eggs and egg-products, tricalcium phosphate and collagen;]
43.
‘processed petfood’ means petfood, other than raw petfood, that has undergone treatment in accordance with the requirements of Annex VIII;
44.
‘processed products’ means animal by-products that have undergone one of the processing methods or another treatment required by Annex VII or VIII;
45.
‘processing methods’ means the methods listed in Annex V, Chapter III;
46.
‘processing plant’ means an animal by-products processing plant;
47.
‘product used for in vitro diagnosis’ means a packaged product, ready for use by the end user, containing a blood product, and used as a reagent, reagent product, calibrator, kit or any other system, whether used alone or in combination, intended to be used in vitro for the examination of samples of human or animal origin, with the exception of donated organs or blood, solely or principally with a view to the diagnosis of a physiological state, state of health, disease or genetic abnormality or to determine safety and compatibility with reagents;
48.
‘raw petfood’ means petfood which has not undergone any preserving process other than chilling, freezing or quick freezing to ensure preservation;
49.
‘remote areas’ means areas where the animal population is so small, and where facilities are so far away, that the arrangements necessary for collection and transport would be unacceptably onerous compared to local disposal;
50.
‘rendered fats’ means fats derived from processing of Category 2 material or Category 3 material;
51.
‘storage plant’ means a plant, other than establishments and intermediaries covered by Directive 95/69/EC(), in which processed products are temporarily stored before their final use or disposal;
52.
‘tanning’ means the hardening of hides, using vegetable tanning agents, chromium salts or other substances such as aluminium salts, ferric salts, silicic salts, aldehydes and quinones, or other synthetic hardening agents;
53.
‘technical plant’ means a plant in which animal by-products are used to produce technical products;
54.
‘technical products’ means products directly derived from certain animal by-products, intended for purposes other than human or animal consumption, including tanned and treated hides and skins, game trophies, processed wool, hair, bristles, feathers and parts of feathers, serum of equidae, blood products, pharmaceuticals, medical devices, cosmetics, bone products for china, gelatin and glue, organic fertilizers, soil improvers, rendered fats, fat derivatives, processed manure and milk and milk-based products;
55.
[‘ unprocessed feathers and parts of feathers ’ means feathers and parts of feathers that have not been treated with a steam current or by some other method that ensures that no pathogens remain;
56.
‘ unprocessed wool ’ means sheep's wool that has not undergone factory washing, been obtained from tanning, or been treated by some other method that ensures that no pathogens remain;
57.
‘ unprocessed hair ’ means ruminant hair that has not undergone factory washing, been obtained from tanning, or been treated by some other method that ensures that no pathogens remain;
58.
‘ unprocessed pig bristles ’ means pig bristles that have not undergone factory washing, been obtained from tanning, or been treated by some other method that ensures that no pathogens remain;]
59.
[‘ collagen ’ means protein-based products derived from hides, skins and tendons of animals, including bones in the case of pigs, poultry and fish;
60.
‘ screenings ’ means visible solid animal materials retained in the waste water screen where a pre-treatment process as referred to in Annex II, Chapter IX, is required;
61.
‘ grease and oil mixture ’ means floating animal materials collected at the surface of waste water grease remover systems where a pre-treatment process as referred to in Annex II, Chapter IX, is required;
62.
‘ sludge ’ means visible solid animal materials or sediments retained in the waste water drains where a pre-treatment process as referred to in Annex II, Chapter IX, is required;
63.
‘material from desanding’ means visible solid animal materials or sediments retained in desanding systems where these constitute a pre-treatment process referred to in Annex II, Chapter IX [;] ]
64.
‘[flavouring innard’ means a liquid or dehydrated processed product of animal origin used to enhance the palatability values of petfood [;]
65.
[‘ colour-coding ’ means the systematic use of colours as defined in Chapter I of Annex II for displaying information as provided for in this Regulation on the surface or part of the surface of a packaging, container or vehicle, or on a label or symbol applied to them.] ]
ANNEX IIU.K.HYGIENE REQUIREMENTS FOR THE COLLECTION AND TRANSPORT OF ANIMAL BY-PRODUCTS AND PROCESSED PRODUCTS
[CHAPTER I U.K. Identification
1. All necessary measures must be taken to ensure that: U.K.
(a)
Category 1, Category 2 and Category 3 materials are identifiable and kept separate and identifiable during collection and transportation;
(b)
processed products are identifiable and kept separate and identifiable during transportation;
(c)
a marking substance for the identification of animal by-products or processed products of a specific category is only used for the category for which its use is required under this Regulation, or is established or laid down pursuant to point 4; and
(d)
animal by-products and processed products are dispatched from one Member State to another Member State in packaging, containers or vehicles which are prominently and, at least for the period of transport, indelibly colour-coded as follows:
(i)
in the case of Category 1 materials, using the colour black;
(ii)
in the case of Category 2 materials (other than manure and digestive tract content), using the colour yellow;
(iii)
in the case of Category 3 materials, using the colour green with a high content of blue to ensure that it is clearly distinguishable from the other colours.
2. During transport, a label attached to the packaging, container or vehicle must: U.K.
(a)
clearly indicate the category of the animal by-products or, in the case of processed products, the category of animal by-products from which the processed products were derived; and
(b)
bear the following words:
(i)
in the case of Category 3 material, ‘ not for human consumption ’ ;
(ii)
in the case of Category 2 material (other than manure and digestive tract content) and processed products derived therefrom, ‘ not for animal consumption ’ ; however, when Category 2 material is intended for the feeding of animals referred to in Article 23(2)(c) under the conditions provided for in that Article, the label shall instead indicate ‘ for feeding to … ’ completed with the name of the specific species of those animals for the feeding of which the material is intended;
(iii)
in the case of Category 1 material and processed products derived therefrom, ‘ for disposal only ’ ;
(iv)
in the case of manure and digestive tract content, ‘ manure ’ .
3. Member States may establish systems or lay down rules for the colour-coding of packaging, containers or vehicles used for the transport of animal by-products and processed products originating in and remaining on their territory, provided that those systems or rules do not confuse the colour-coding system provided for in point 1(d). U.K.
4. Without prejudice to point 3 of Annex V to Regulation (EC) No 999/2001, Member States may establish systems or lay down rules for the marking of animal by-products originating in and remaining on their territory provided that those systems or rules do not conflict with the marking requirements laid down for processed products in Chapter I of Annex VI to this Regulation. U.K.
5. By way of derogation from points 3 and 4, Member States may use the systems or rules referred to in those points for animal by-products originating in but not intended to remain on their territory if the Member State or third country of destination has communicated its agreement.] U.K.
CHAPTER IIU.K.Vehicles and containers
1.Animal by-products and processed products must be collected and transported in sealed new packaging or covered leak-proof containers or vehicles.U.K.
2.Vehicles and reusable containers, and all reusable items of equipment or appliances that come into contact with animal by-products or processed products, must be:U.K.
(a)
cleaned, washed and disinfected after each use;
(b)
maintained in a clean condition; and
(c)
clean and dry before use.
3.Reusable containers must be dedicated to the carriage of a particular product to the extent necessary to avoid cross-contamination.U.K.
[4. Packaging material must be incinerated or disposed of by some other means in accordance with instructions from the competent authority.] U.K.
CHAPTER IIIU.K.Commercial documents and health certificates
[1. During transportation, a commercial document or, when required by this Regulation, a health certificate must accompany animal by-products and processed products except in the case of processed products originating from Category 3 material which are supplied within the same Member State by retailers to final users other than business operators.] U.K.
2.Commercial documents must specify:U.K.
(a)
the date on which the material was taken from the premises;
(b)
the description of the material, including the information referred to in Chapter I, the animal species for Category 3 material and processed products derived therefrom destined for use as feed material and, if applicable, the ear-tag number;
(c)
the quantity of the material;
(d)
the place of origin of the material;
(e)
the name and the address of the carrier;
(f)
the name and the address of the receiver and, if applicable, its approval number; and
(g)
if appropriate:
(i)
the approval number of the plant of origin, and
(ii)
the nature and the methods of the treatment.
3.The commercial document must be produced at least in triplicate (one original and two copies). The original must accompany the consignment to its final destination. The receiver must retain it. The producer must retain one of the copies and the carrier the other.U.K.
4.A model for the commercial document may be laid down under the procedure referred to in Article 33(2).U.K.
5.Health certificates must be issued and signed by the competent authority.U.K.
CHAPTER IVU.K.Records
The records referred to in Article 9 must contain the information referred to in Chapter III, paragraph 2, as follows. They must contain:
(a)
the information referred to in subparagraphs (b) and (c); and
(b)
in the case of records kept by any person consigning animal by-products, the information referred to in subparagraphs (a), (e) and, if known, (f); or
(c)
in the case of records kept by any person transporting animal by-products, the information referred to in subparagraphs (a), (d) and (f); or
(d)
in the case of records kept by any person receiving animal by-products, the date of reception and the information referred to in subparagraphs (d) and (e).
CHAPTER VU.K.Retention of documents
The commercial document and the health certificate referred to in Chapter III, and the records referred to in Chapter IV, must be kept for a period of at least two years for presentation to the competent authority.
CHAPTER VIU.K.Temperature conditions
1.The transport of animal by-products must take place at an appropriate temperature, to avoid any risk to animal or public health.U.K.
2.Unprocessed Category 3 material destined for the production of feed material or pet food must be transported chilled or frozen, unless processed within 24 hours of departure.U.K.
3.The design of vehicles used for refrigerated transport must ensure the maintenance of an appropriate temperature throughout transport.U.K.
CHAPTER VIIU.K.Specific rules for transit
The carriage of animal by-products and processed products in transit must meet the requirements of Chapters I, II, III and VI.
CHAPTER VIIIU.K.Control measures
The competent authority must take the necessary measures to control the collection, transport, use and disposal of animal by-products and processed products, including by checking the keeping of required records and documents and, when this Regulation requires it or the competent authority considers it necessary, by sealing.
When the competent authority applies a seal to a consignment of animal by-products or processed products, it must inform the competent authority of the place of destination.
[CHAPTER IX U.K. Collection of animal material when treating waste water
1. Category 1 processing plants and other premises where specified risk material is removed, slaughterhouses and Category 2 processing plants shall have a pre-treatment process for the retention and collection of animal material as an initial step in the treatment of waste water. The equipment used in the pre-treatment process shall consist of drain traps or screen with apertures or a mesh size of no more than 6 mm in the downstream end of the process or equivalent systems that ensures that the solid particles in the waste water passing through them are no more than 6 mm. U.K.
2. Waste water from the premises as referred to in paragraph 1 must enter a pre-treatment process which shall ensure that all waste water has been filtered through the process before being drained off the premises. No grinding or maceration shall take place which could facilitate the passage of animal material through the pre-treatment process. U.K.
3. All animal material retained in the pre-treatment process in premises as referred to in paragraph 1 shall be collected and transported as Category 1 or Category 2 material, as appropriate, and disposed of in accordance with this Regulation. U.K.
4. Waste water having passed the pre-treatment process in premises referred to in paragraph 1 and waste water from premises only receiving Category 3 material shall be treated in accordance with other relevant Community legislation.] U.K.
[CHAPTER X U.K. Commercial document
1.
[A commercial document in accordance with the model set out in this Chapter shall accompany animal by-products and processed products during transportation. However, for the transport of animal by-products and processed products on their own territory Member States may require:
(a)
to use a different commercial document, in paper or in electronic form, provided that such commercial document complies with the requirements laid down in point 2 of Chapter III;
(b)
that the quantity of the material referred to in point 2(c) of Chapter III is expressed in weight of the material in the commercial document;
(c)
that a copy of the commercial document is returned by the receiver to the producer to be kept by that producer in accordance with Chapter V as proof of arrival of the consignment.]
2.
Where more than one transporter is involved, each transporter shall fill in a declaration as referred to in point 7 of the commercial document, which shall be part of the document.
MODEL COMMERCIAL DOCUMENT FOR THE TRANSPORTATION WITHIN THE EUROPEAN COMMUNITY OF ANIMAL BY-PRODUCTS AND PROCESSED PRODUCTS U.K.
Notes U.K.
(a) Commercial documents shall be produced, according to the layout of the model appearing in this Annex. It shall contain, in the numbered order that appears in the model, the attestations that are required for the transportation of animal by-products and processed products derived there from. U.K.
(b) It shall be drawn up in one of the official languages of the MemberState of origin or the MemberState of destination, as appropriate. However, it may also be drawn up in other official Community languages, if accompanied by an official translation or if previously agreed by the competent authority of the MemberState of destination. U.K.
(c) The commercial document must be produced at least in triplicate (one original document and two copies). The original document must accompany the consignment to its final destination. The receiver must retain it. The producer must retain one of the copies and the carrier the other. U.K.
(d) The original of each commercial document shall consist of a single page, both sides, or, where more text is required it shall be in such a form that all pages needed are part of an integrated whole and indivisible. U.K.
(e) If for reasons of identification of the items of the consignment, additional pages are attached to the commercial document, these pages shall also be considered as forming part of the original document by the application of the signature of the person responsible for the consignment, on each of the pages. U.K.
(f) When the commercial document, including additional pages referred to in (e), comprises more than one page, each page shall be numbered — ( page number ) of ( total number of pages ) — at the bottom and shall bear the code number of the document that has been designated by the responsible person at the top. U.K.
(g) The original of the commercial document must be completed and signed by the responsible person. In doing so, the responsible person shall ensure that the principles of documentation as laid down in Chapter III of Annex II to Regulation (EC) No 1774/2002 are followed. The commercial document must specify: U.K.
(i)
the date on which the material was taken from the premises;
(ii)
the description of the material, including the identification of the material, the animal species for Category 3 material and processed products derived therefrom destined for use as feed material and, if applicable, the ear-tag number of the animal;
(iii)
the quantity of the material;
(iv)
the place of origin of the material;
(v)
the name and the address of the carrier of the material;
(vi)
the name and the address of the receiver and, if applicable, its approval number; and
(vii)
if appropriate, the approval number of the plant of origin, and the nature and the methods of the treatment.
(h) The colour of the signature of the responsible person shall be different to that of the printing. U.K.
(i) The commercial document must be kept for a period of at least two years for presentation to the competent authority to verify the records referred to in Article 9 of Regulation (EC) No 1774/2002. U.K.
(j) Where Member States decide to use a commercial document in electronic form, the requirements listed in points (a) to (i) shall be complied with as appropriate for such electronic form.] U.K.



ANNEX IIIU.K.HYGIENE REQUIREMENTS FOR INTERMEDIATE AND STORAGE PLANTS
CHAPTER IU.K.Requirements for the approval of intermediate plants
1.Premises and facilities must meet at least the following requirements.U.K.
(a)
The premises must be adequately separated from the public highway and other premises such as slaughterhouses. The layout of plants must ensure the total separation of Category 1 and Category 2 material from Category 3 material from reception until dispatch.
(b)
The plant must have a covered space to receive animal by-products.
(c)
The plant must be constructed in such a way that it is easy to clean and disinfect. Floors must be laid down in such a way as to facilitate the draining of liquids.
(d)
The plant must have adequate lavatories, changing rooms and washbasins for staff.
(e)
The plant must have appropriate arrangements for protection against pests, such as insects, rodents and birds.
(f)
The plant must have a waste-water disposal system which meets hygiene requirements.
(g)
Where it is necessary for the purpose of achieving the objectives of this Regulation, plants must have suitable temperature-controlled storage facilities of sufficient capacity for maintaining animal by-products at appropriate temperatures and designed to allow the monitoring and recording of those temperatures.
2.The plant must have adequate facilities for cleaning and disinfecting the containers or receptacles in which animal by-products are received and the vehicles, other than ships, in which they are transported. Adequate facilities must be provided for the disinfecting of vehicle wheels.U.K.
CHAPTER IIU.K.General hygiene requirements
A.Category 3 intermediate plantsU.K.
1.The plant must not engage in activities other than the importation, collection, sorting, cutting, chilling, freezing into blocks, temporary storage and dispatching of Category 3 material.U.K.
2.The sorting of Category 3 material must be carried out in such a way as to avoid any risk of the propagation of animal diseases.U.K.
3.All the time during sorting or storage, Category 3 material must be handled and stored separately from goods other than other Category 3 material and in such a way as to prevent any propagation of pathogens and to ensure compliance with Article 22.U.K.
4.Category 3 material must be stored properly, and, where appropriate, chilled or frozen, until re-dispatched.U.K.
5.. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .U.K.
B.Category 1 or Category 2 intermediate plantsU.K.
6.The plant must not engage in activities other than the collection, handling, temporary storage and dispatching of Category 1 or Category 2 material.U.K.
7.The sorting of the Category 1 or Category 2 material must be carried out in such a way as to avoid any risk of the propagation of animal diseases.U.K.
8.All the time during storage, the Category 1 or Category 2 material must be handled and stored separately from other goods and in such a way as to prevent any propagation of pathogens.U.K.
9.Category 1 or Category 2 material must be stored properly, including under appropriate temperature conditions, until re-dispatched.U.K.
10.. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .U.K.
[11. Waste water must be treated to ensure, as far as is reasonably practicable, that no pathogens remain. Specific requirements for the treatment of waste water from Category 1 and Category 2 intermediate plants may be laid down by the Commission. Those measures, designed to amend non-essential elements of this Regulation by supplementing it, shall be adopted in accordance with the regulatory procedure with scrutiny referred to in Article 33(3).] U.K.
CHAPTER IIIU.K.Requirements for the approval of storage plants
Premises and facilities must meet at least the following requirements.
1.
Premises storing processed products derived from Category 3 material must not be at the same site as premises storing processed products derived from Category 1 or Category 2 material, unless in a completely separate building.
2.
The plant must:
(a)
have a covered space to receive the products;
(b)
be constructed in such a way that it is easy to clean and disinfect. Floors must be laid down in such a way as to facilitate the draining of liquids;
(c)
have adequate lavatories, changing rooms and washbasins for staff; and
(d)
have appropriate arrangements for protection against pests, such as insects, rodents and birds.
3.
The plant must have adequate facilities for cleaning and disinfecting the containers or receptacles in which the products are received and the vehicles, other than ships, in which they are transported. Adequate facilities must be provided for the disinfecting of vehicle wheels.
4.
Products must be stored properly until re-dispatched.
ANNEX IVU.K.REQUIREMENTS FOR INCINERATION AND CO-INCINERATION PLANTS TO WHICH DIRECTIVE 2000/76/EC DOES NOT APPLY
CHAPTER IU.K.General conditions
[1. Incineration or co-incineration plants must be designed, equipped and operated in such a manner as to fulfil the requirements of this Regulation. The following hygiene conditions must be met: U.K.
(a)
Animal by-products must be disposed of as soon as possible after arrival. They must be stored properly until disposal.
(b)
Containers, receptacles and vehicles used for transporting unprocessed material must be cleaned in a designated area, thereby ensuring that waste water is treated during the storage referred to in Chapter III.
(c)
Preventive measures against birds, rodents, insects or other vermin must be taken systematically. A documented pest control programme must be used for that purpose.
(d)
Cleaning procedures must be established and documented for all parts of the premises. Suitable equipment and cleaning agents must be provided for cleaning.
(e)
Hygiene control must include regular inspections of the environment and equipment. Inspection schedules and results must be documented and maintained for at least two years.]
2.The operator of an incineration or co-incineration plant must take all necessary precautions concerning the reception of animal by-products to prevent, or limit as far as practicable, direct risks to human or animal health.U.K.
CHAPTER IIU.K.Operating conditions
3.Incineration or co-incineration plants must be designed, equipped, built and operated in such a way that the gas resulting from the process is raised in a controlled and homogeneous fashion, even under the most unfavourable conditions, to a temperature of 850 oC, as measured near the inner wall or at another representative point of the combustion chamber as authorised by the competent authority, for two seconds.U.K.
4.Each line of high-capacity incineration plants must be equipped with at least one auxiliary burner. This burner must be switched on automatically when the temperature of the combustion gases after the last injection of combustion air falls below 850 oC. It must also be used during plant start-up and shut-down operations to ensure that the temperature of 850 oC is maintained at all times during these operations and as long as unburned material is in the combustion chamber.U.K.
5.High-capacity incineration or co-incineration plants must have and operate an automatic system to prevent feed with animal by-products:U.K.
(a)
at start-up, until the temperature of 850 oC has been reached; and
(b)
whenever the temperature of 850 oC is not maintained.
6.Animal by-products should, where practicable, be placed straight in the furnace without direct handling.U.K.
CHAPTER IIIU.K.Water discharges
7.Incineration or co-incineration plant sites, including associated storage areas for animal by-products, must be designed in such a way as to prevent unauthorised and accidental release of any polluting substances into soil, surface water and groundwater in accordance with the provisions provided for in relevant Community legislation. Moreover, storage capacity must be provided for contaminated rainwater run-off from the incineration plant site or for contaminated water arising from spillage or fire-fighting operations.U.K.
8.The storage capacity must be adequate to ensure that such waters can be tested and treated before discharge where necessary.U.K.
CHAPTER IVU.K.Residues
9.For the purposes of this Chapter, ‘residues’ means any liquid or solid material generated by the incineration or co-incineration process, the waste-water treatment or other processes within the incineration or co-incineration plant. They include bottom ash and slag, fly ash and boiler dust.U.K.
10.Residues resulting from the operation of the incineration or co-incineration plant must be minimised in their amount and harmfulness. Residues must be recycled, where appropriate, directly in the plant or outside in accordance with relevant Community legislation.U.K.
11.Transport and intermediate storage of dry residues in the form of dust must take place in such a way as to prevent dispersal in the environment (e.g., in closed containers).U.K.
CHAPTER VU.K.Temperature measurement
12.Techniques must be used to monitor the parameters and conditions relevant to the incineration or co-incineration process. High-capacity incineration and co-incineration plants must have and use temperature measurement equipment.U.K.
13.The approval issued by the competent authority, or conditions attached to it, must lay down temperature measurement requirements.U.K.
14.The appropriate installation and the functioning of any automated monitoring equipment must be subject to control and to an annual surveillance test. Calibration must be carried out by means of parallel measurements with the reference methods at least every three years.U.K.
15.Temperature measurement results must be recorded and presented in an appropriate fashion to enable the competent authority to verify compliance with the permitted operating conditions laid down in this Regulation in accordance with procedures to be decided upon by that authority.U.K.
CHAPTER VIU.K.Abnormal operating
16.In the case of a breakdown, or abnormal operating conditions, the operator must reduce or close down operations as soon as practicable until normal operations can be resumed.U.K.
[CHAPTER VII U.K. Incineration of Category 1 material referred to in Article 4(1)(b)
1. The low-capacity incineration plant must be located on a well-drained hard standing. U.K.
2. Livestock must not have access to the low-capacity incineration plant, animal by-products that are awaiting incineration or ash resulting from the incineration of animal by-products. If the low-capacity incineration plant is located on a livestock holding: U.K.
(a)
there must be total physical separation between the incinerator and the livestock and their feed and bedding, with fencing where necessary;
(b)
equipment must be dedicated entirely to the operation of the incinerator and not used elsewhere on the farm;
(c)
the operators must change their outer clothing and footwear before handling livestock or livestock feed.
3. The storage of animal by-products and of ashes must be covered, labelled and leak proof. U.K.
4. The operator must check that animal by-products are incinerated in such a way that they are completely reduced to ash. Ash must be disposed of to a landfill approved under Directive 1999/31/EC. U.K.
5. Incompletely incinerated animal by-products must not be disposed of to a landfill, but must be re-incinerated or otherwise disposed of in accordance with this Regulation. U.K.
6. The low-capacity incineration plant must be equipped with an afterburner. U.K.
7. The operator must keep records of the quantities, category and species of animal by-products incinerated and the date of incineration. U.K.
8. The competent authority must inspect the low-capacity incineration plant before approval, and at least once a year to monitor compliance with this Regulation.] U.K.
ANNEX VU.K.GENERAL HYGIENE REQUIREMENTS FOR THE PROCESSING OF CATEGORY 1, 2 AND 3 MATERIAL
CHAPTER IU.K.General requirements for the approval of Category 1, 2 and 3 processing plants
1.Premises and facilities must meet at least the following requirements:U.K.
(a)
[Processing plants shall not be situated on the same site as slaughterhouses, unless the risks to public and animal health, resulting from the processing of animal by-products which originate from such slaughterhouses, are mitigated by compliance with at least the following conditions:
(i)
the processing plant must be physically separated from the slaughterhouse; where appropriate by locating the processing plant in a building that is completely separated from the slaughterhouse;
(ii)
the following must be installed and operated:
a conveyer system which links the processing plant to the slaughterhouse,
separate entrances, reception bays, equipment and exits for the processing plant and the slaughterhouse;
(iii)
measures must be taken to prevent the spreading of risks through the operation of personnel which is employed in the processing plant and in the slaughterhouse;
(iv)
unauthorised persons and animals must not have access to the processing plant.
By way of derogation from points (i) to (iv), in the case of Category 3 processing plants, the competent authority may authorise other conditions instead of those set out in those points, aimed at mitigating the risks to public and animal health, including the risks arising from the processing of Category 3 material, which originates from off-site establishments approved under Regulation (EC) No 853/2004. Member States shall inform the Commission and the other Member States in the framework of the Committee referred to in Article 33(1) of the use made of this derogation by their competent authorities;]
(b)
the processing plant must have a clean and unclean sector, adequately separated. The unclean sector must have a covered place to receive animal by-products and must be constructed in such a way that it is easy to clean and disinfect. Floors must be laid in such a way as to facilitate the draining of liquids. The processing plant must have adequate lavatories, changing rooms and washbasins for staff;
(c)
the processing plant must have sufficient production capacity for hot water and steam for the processing of animal by-products;
(d)
the unclean sector must, if appropriate, contain equipment to reduce the size of animal by-products and equipment for loading the crushed animal by-products into the processing unit;
(e)
all installations in which animal by-products are processed must operate in accordance with the requirements of Chapter II. Where heat treatment is required, all installations must be equipped with:
(i)
measuring equipment to monitor temperature against time and, if necessary, pressure at critical points;
(ii)
recording devices to record continuously the results of these measurements; and
(iii)
an adequate safety system to prevent insufficient heating;
(f)
to prevent recontamination of the finished product by incoming animal by-products, there must be a clear separation between the area of the plant where incoming material for processing is unloaded and the areas set aside for the processing of that product and the storage of the processed product.
2.The processing plant must have adequate facilities for cleaning and disinfecting the containers or receptacles in which animal by-products are received and the vehicles, other than ships, in which they are transported.U.K.
3.Adequate facilities must be provided for the disinfecting of vehicle wheels, on leaving the unclean sector of the processing plant.U.K.
4.All processing plants must have a waste-water disposal system meeting the competent authority's requirements.U.K.
5.The processing plant must have its own laboratory or make use of the services of an external laboratory. The laboratory must be equipped to carry out necessary analyses and be approved by the competent authority.U.K.
CHAPTER IIU.K.General hygiene requirements
1.Animal by-products must be processed as soon as possible after arrival. They must be stored properly until processed.U.K.
2.Containers, receptacles and vehicles used for transporting unprocessed material must be cleaned in a designated area. That area must be situated or designed to prevent the risk of contamination of processed products.U.K.
3.Persons working in the unclean sector must not enter the clean sector without changing their working clothes and footwear or without disinfecting the latter. Equipment and utensils must not be taken from the unclean sector into the clean sector, unless first cleaned and disinfected. Personnel movement procedures must be established to control the movement of personnel between areas and to prescribe the proper use of foot baths and wheel baths.U.K.
[4. Waste water originating in the unclean sector must be treated to ensure, as far as is reasonably practicable, that no pathogens remain. Specific requirements for the treatment of waste water from processing plants may be laid down by the Commission. Those measures, designed to amend non-essential elements of this Regulation by supplementing it, shall be adopted in accordance with the regulatory procedure with scrutiny referred to in Article 33(3).] U.K.
5.Preventive measures against birds, rodents, insects or other vermin must be taken systematically. A documented pest control programme must be used for that purpose.U.K.
6.Cleaning procedures must be established and documented for all parts of the premises. Suitable equipment and cleaning agents must be provided for cleaning.U.K.
7.Hygiene control must include regular inspections of the environment and equipment. Inspection schedules and results must be documented and maintained for at least two years.U.K.
8.Installations and equipment must be kept in a good state of repair and measuring equipment must be calibrated at regular intervals.U.K.
9.Processed products must be handled and stored at the processing plant in such a way as to preclude recontamination.U.K.
CHAPTER IIIU.K.Processing methods
Method 1U.K.
ReductionU.K.
1.If the particle size of the animal by-products to be processed is more than 50 millimetres, the animal by-products must be reduced in size using appropriate equipment, set so that the particle size after reduction is no greater than 50 millimetres. The effectiveness of the equipment must be checked daily and its condition recorded. If checks disclose the existence of particles larger than 50 millimetres, the process must be stopped and repairs made before the process is resumed.U.K.
Time, temperature and pressureU.K.
2.After reduction the animal by-products must be heated to a core temperature of more than 133 oC for at least 20 minutes without interruption at a pressure (absolute) of at least 3 bars produced by saturated steam(); the heat treatment may be applied as the sole process or as a pre- or post-process sterilisation phase.U.K.
3.The processing may be carried out in batch or continuous systems.U.K.
Method 2U.K.
ReductionU.K.
1.If the particle size of the animal by-products to be processed is more than 150 millimetres, the animal by-products must be reduced in size using appropriate equipment, set so that the particle size after reduction is no greater than 150 millimetres. The effectiveness of the equipment must be checked daily and its condition recorded. If checks disclose the existence of particles larger than 150 millimetres, the process must be stopped and repairs made before the process is resumed.U.K.
Time, temperature and pressureU.K.
2.After reduction the animal by-products must be heated to a core temperature greater than 100 oC for at least 125 minutes, a core temperature greater than 110 oC for at least 120 minutes and a core temperature greater that 120 oC for at least 50 minutes.U.K.
3.The processing must be carried out in a batch system.U.K.
[4. The animal by-products may be cooked in such a manner that the time-temperature requirements are achieved at the same time.] U.K.
Method 3U.K.
ReductionU.K.
1.If the particle size of the animal by-products to be processed is more than 30 millimetres, the animal by-products must be reduced in size using appropriate equipment, set so that the particle size after reduction is no greater than 30 millimetres. The effectiveness of the equipment must be checked daily and its condition recorded. If checks disclose the existence of particles larger than 30 millimetres, the process must be stopped and repairs made before the process is resumed.U.K.
Time, temperature and pressureU.K.
2.After reduction the animal by-products must be heated to a core temperature greater than 100 oC for at least 95 minutes, a core temperature greater than 110 oC for at least 55 minutes and a core temperature greater that 120 oC for at least 13 minutes.U.K.
3.The processing may be carried out in batch or continuous systems.U.K.
4.The animal by-products may be cooked in such a manner that the time-temperature requirements are achieved at the same time.U.K.
Method 4U.K.
ReductionU.K.
1.If the particle size of the animal by-products to be processed is more than 30 millimetres, the animal by-products must be reduced in size using appropriate equipment, set so that the particle size after reduction is no greater than 30 millimetres. The effectiveness of the equipment must be checked daily and its condition recorded. If checks disclose the existence of particles larger than 30 millimetres, the process must be stopped and repairs made before the process is resumed.U.K.
Time, temperature and pressureU.K.
2.After reduction the animal by-products must be placed in a vessel with added fat and heated to a core temperature greater than 100 oC for at least 16 minutes, a core temperature greater than 110 oC for at least 13 minutes, a core temperature greater that 120 oC for at least eight minutes and a core temperature greater that 130 oC for at least three minutes.U.K.
3.The processing may be carried out in batch or continuous systems.U.K.
4.The animal by-products may be cooked in such a manner that the time-temperature requirements are achieved at the same time.U.K.
Method 5U.K.
ReductionU.K.
1.If the particle size of the animal by-products to be processed is more than 20 millimetres, the animal by-products must be reduced in size using appropriate equipment, set so that the particle size after reduction is no greater than 20 millimetres. The effectiveness of the equipment must be checked daily and its condition recorded. If checks disclose the existence of particles larger than 20 millimetres, the process must be stopped and repairs made before the process is resumed.U.K.
Time, temperature and pressureU.K.
2.After reduction the animal by-products must be heated until they coagulate and then pressed so that fat and water are removed from the proteinaceous material. The proteinaceous material must then be heated to a core temperature greater than 80 oC for at least 120 minutes and a core temperature greater than 100 oC for at least 60 minutes.U.K.
3.The processing may be carried out in batch or continuous systems.U.K.
4.The animal by-products may be cooked in such a manner that the time-temperature requirements are achieved at the same time.U.K.
[Method 6 (For Category 3 animal by-products of fish origin only) U.K.
Reduction U.K.
1. The animal by-products must be reduced to at least: U.K.
(a)
50 mm in case of heat treatment in accordance with paragraph 2(a); or
(b)
30 mm in case of heat treatment in accordance with paragraph 2(b).
They must then be mixed with formic acid to reduce and maintain the pH to 4,0 or lower. The mixture must be stored for at least 24 hours pending further treatment.
Time and temperature U.K.
2. Following reduction, the mixture must be heated to: U.K.
(a)
a core temperature of at least 90 °C for at least 60 minutes; or
(b)
a core temperature of at least 70 °C for at least 60 minutes.
When using a continuous flow system, the progression of the product through the heat converter must be controlled by means of mechanical commands limiting its displacement in such a way that at the end of the heat treatment operation the product has undergone a cycle which is sufficient in both time and temperature.]
Method 7U.K.
1.Any processing method approved by the competent authority where it has been demonstrated to that authority that the final product has been sampled on a daily basis over a period of one month in compliance with the following microbiological standards:U.K.
(a)
Samples of material taken directly after heat treatment:
(b)
Samples of material taken during or upon withdrawal from storage at the processing plant:
Salmonella: absence in 25 g: n = 5, c = 0, m = 0, M = 0
Enterobacteriaceae: n = 5, c = 2, m = 10, M = 300 in 1 g
where:
n
=
number of samples to be tested;
m
=
threshold value for the number of bacteria; the result is considered satisfactory if the number of bacteria in all samples does not exceed m;
M
=
maximum value for the number of bacteria; the result is considered unsatisfactory if the number of bacteria in one or more samples is M or more; and
c
=
number of samples the bacterial count of which may be between m and M, the sample still being considered acceptable if the bacterial count of the other samples is m or less.
2.Details of the critical control points under which each processing plant satisfactorily complies with the microbiological standards must be recorded and maintained so that the owner, operator or their representative and the competent authority can monitor the operation of the processing plant. The information to be recorded and monitored must include the particle size, critical temperature and, as appropriate, the absolute time, pressure profile, raw material feed-rate and fat recycling rate.U.K.
3.This information must be made available to the Commission on request.U.K.
CHAPTER IVU.K.Supervision of production
1.The competent authority must supervise processing plants to ensure compliance with the requirements of this Regulation. It must in particular:U.K.
(a)
check:
(i)
the general conditions of hygiene of the premises, equipment and staff;
(ii)
the efficacy of the own checks carried out by the plant, in accordance with Article 25, particularly by examining the results and taking samples;
(iii)
the standards of the products after processing. The analyses and tests must be carried out in accordance with scientifically-recognised methods (in particular, those laid down in Community legislation or, where none exist, recognised international standards or, in their absence, national standards); and
(iv)
the storage conditions;
(b)
take any samples required for laboratory tests; and
(c)
make any other checks it considers necessary to ensure compliance with this Regulation.
2.To allow it to carry out its responsibilities under paragraph 1, the competent authority must have free access at all times to all parts of the processing plant and to records, commercial documents and health certificates.U.K.
CHAPTER VU.K.Validation procedures
1.The competent authority must validate the processing plant in accordance with the following procedures and indicators:U.K.
(a)
description of the process (by a process flow diagram);
(b)
identification of critical control points (CCPs) including the material process rate for continuous systems;
(c)
compliance with the specific process requirements laid down by this Regulation; and
(d)
achievement of the following requirements:
(i)
particle size for batch-pressure and continuous processes — defined by the mincer hole or the anvil gap size, and
(ii)
temperature, pressure, processing time and material processing rate (for continuous system only) as specified in paragraphs 2 and 3.
2.In the case of a batch pressure system:U.K.
(a)
the temperature must be monitored with a permanent thermocouple and it must be plotted against real time;
(b)
the pressure stage must be monitored with a permanent pressure gauge. Pressure must be plotted against real time;
(c)
the processing time must be shown by time/temperature and time/pressure diagrams.
At least once a year the thermocouple and the pressure gauge must be calibrated.
3.In the case of a continuous pressure system:U.K.
(a)
the temperature and the pressure must be monitored with thermocouples, or an infrared temperature gun, and pressure gauges used at defined positions throughout the process system in such a way that temperature and pressure comply with the required conditions inside the whole continuous system or in a section of it. The temperature and pressure must be plotted against real time;
(b)
measurement of the minimum transit time inside the whole relevant part of the continuous system where the temperature and pressure comply with the required conditions, must be provided to the competent authorities, using insoluble markers (for example, manganese dioxide) or a method which offers equivalent guarantees. Accurate measurement and control of the material process rate is essential and must be measured during the validation test in relation to a CCP that can be continuously monitored such as:
(i)
feed screw revolutions per minute (rev./min.),
(ii)
electric power (amps at given voltage),
(iii)
evaporation/condensation rate, or
(iv)
number of pump strokes per unit time.
All measuring and monitoring equipment must be calibrated at least once a year.
4.The competent authority must repeat the validation procedures periodically, when it considers it necessary, and in any case each time any significant alterations are made to the process (for example, modification of the machinery or a change of raw materials).U.K.
[5. Validation procedures based on testing methods may be laid down by the Commission. Those measures, designed to amend non-essential elements of this Regulation by supplementing it, shall be adopted in accordance with the regulatory procedure with scrutiny referred to in Article 33(3).] U.K.
ANNEX VIU.K. [SPECIFIC REQUIREMENTS FOR THE PROCESSING OF CATEGORY 1 AND 2 MATERIAL, FOR BIOGAS AND COMPOSTING AND FOR THE MARKING OF CERTAIN PROCESSED PRODUCTS]
CHAPTER IU.K. [Specific requirements for the processing of Category 1 and 2 materials and for the marking of certain processed products]
The following requirements apply in addition to the general requirements laid down in Annex V.
A.
Premises
1.
The layout of Category 1 and Category 2 processing plants must ensure the total separation of Category 1 material from Category 2 material from reception of the raw material until dispatch of the resulting processed product.
2.
However, the competent authority may authorise the temporary use of a Category 2 processing plant for the processing of Category 1 material when a widespread outbreak of an epizootic disease or other extraordinary and unforeseeable circumstances leads to a lack of capacity at a Category 1 processing plant.
The competent authority must re-approve the Category 2 processing plant in accordance with Article 13 before it processes Category 2 material again.
B.
Processing standards
3.
The critical control points that determine the extent of the heat treatments applied in processing must be identified for each processing method as specified in Annex V, Chapter III. The critical control points may include:
(a)
raw material particle size;
(b)
temperature achieved in the heat treatment process;
(c)
pressure applied to the raw material; and
(d)
duration of the heat treatment process or feed rate to a continuous system.
Minimum process standards must be specified for each applicable critical control point.
4.
Records must be maintained for at least two years to show that the minimum process values for each critical control point are applied.
5.
Accurately calibrated gauges/recorders must be used to monitor continuously the processing conditions. Records must be kept to show the date of calibration of gauges/recorders.
6.
Material that may not have received the specified heat treatment (e.g. material discharged at start up, or leakage from cookers) must be recirculated through the heat treatment or collected and reprocessed.
7.
Animal by-products must be processed in accordance with the following processing standards.
(a)
Processing method 1 must be applied to:
(i)
[Category 2 material (other than manure, digestive tract content separated from the digestive tract, milk and colostrum), destined for biogas or composting plants or intended to be used as organic fertilisers or soil improvers, and]
(ii)
Category 1 and Category 2 material destined for landfill.
(b)
Any of processing methods 1 to 5 must be applied to:
(i)
Category 2 material from which the resulting protein is destined for incineration or co-incineration,
(ii)
Category 2 material from which the rendered fat is destined for a Category 2 oleochemical plant, and
(iii)
Category 1 or Category 2 material destined for incineration or co-incineration.
[ . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .]
C.
Processed products
8.
[Processed products derived from Category 1 or 2 material, with the exception of liquid products destined for biogas or composting plants, must be permanently marked, where technically possible with smell, using a system approved by the competent authority. Detailed rules for such marking may be laid down by the Commission. Those measures, designed to amend non-essential elements of this Regulation by supplementing it, shall be adopted in accordance with the regulatory procedure with scrutiny referred to in Article 33(3).]
9.
Samples of processed products destined for biogas or composting plants or landfill, taken directly after heat treatment, must be free from heat-resistant pathogenic bacteria spores (Clostridium perfringens absent in 1 g of the products).
10.
[In processing plants approved in accordance with Article 13, processed products as referred to in Article 4(2)(b) and (c) and Article 5(2)(b) and (c) shall be permanently marked with:
(a)
smell, where technically possible; and
(b)
glyceroltriheptanoate (GTH) in such a way that:
(i)
GTH is added to processed products that have undergone a preceding sanitising thermal treatment at a core temperature of at least 80 °C and remain subsequently protected from re-contamination; and
(ii)
all processed products contain homogenously throughout the substance a minimum concentration of at least 250 mg GTH per kg fat.
11.
The operators of processing plants approved in accordance with Article 13 shall have in place a system of constant monitoring and recording of parameters suitable to demonstrate to the competent authority that the required homogeneous minimum concentration of GTH as referred to in point 10(b) is achieved in the processed products referred to in point 10.
That monitoring and recording system shall include the determination of the content of intact GTH as triglyceride in a cleaned petroleum-ether 40-70 extract of GTH from samples taken at regular intervals.
12.
The competent authority shall carry out a performance check of the monitoring and recording system referred to in point 11 to ascertain compliance with this Regulation and may, where necessary, request the testing of additional samples in accordance with the method referred to in the second paragraph of point 11.
13.
The marking with GTH shall not be required for processed products as referred to in Article 4(2)(b) and (c) and Article 5(2)(b) and (c), where such products are:
(a)
moved by a closed conveyer system, where such a system has been authorised by the competent authority, from the processing plant for:
(i)
immediate direct incineration or co-incineration; or
(ii)
immediate use in accordance with a method approved for Category 1 and 2 animal by-products in accordance with Articles 1 and 2 of Regulation (EC) No 92/2005; or
(b)
intended for research or for scientific use authorised by the competent authority.]
CHAPTER IIU.K.Specific requirements for the approval of biogas and composting plants
A.
Premises
1.
[A biogas plant must be equipped with:
(a)
a pasteurisation/hygienisation unit, which cannot be by-passed, with:
(i)
installations for monitoring temperature against time;
(ii)
recording devices to record continuously the results of the monitoring measurements referred to in (i); and
(iii)
an adequate safety system to prevent insufficient heating;
(b)
adequate facilities for the cleaning and disinfecting of vehicles and containers upon leaving the biogas plant.
However, a pasteurisation/hygienisation unit shall not be mandatory for biogas plants that transform only:
(i)
animal by-products that have undergone processing Method 1;
(ii)
Category 3 material that has undergone pasteurisation/hygienisation elsewhere; or
(iii)
animal by-products which may be used as raw material without processing.
If the biogas plant is located on premises where farmed animals are kept and does not only use manure which accrues from those animals, the plant shall be located at an adequate distance from the area where such animals are kept and there must be, in any case, total physical separation between that plant and those animals and their feed and bedding, with fencing where necessary.
2.
A composting plant must be equipped with:
(a)
a closed composting reactor, which cannot be by-passed, with:
(i)
installations for monitoring temperature against time;
(ii)
recording devices to record, where appropriate continuously, the results of the monitoring measurements referred to in (i); and
(iii)
an adequate safety system to prevent insufficient heating;
(b)
adequate facilities for cleaning and disinfecting of vehicles and containers transporting untreated animal by-products.
However, other types of composting systems may be allowed provided they:
(i)
ensure adequate measures to control vermin;
(ii)
are managed in such a way that all the material in the system achieves the required time and temperature parameters, including, where appropriate, continuous monitoring of the parameters;
(iii)
comply with all other requirements of this Regulation.
If the composting plant is located on premises where farmed animals are kept and does not only use manure which accrues from those animals, the composting plant shall be located at an adequate distance from the area where animals are kept and there must, in any case, be total physical separation between that composting plant and the animals and their feed and bedding, with fencing where necessary.]
3.
Each biogas plant and composting plant must have its own laboratory or make use of an external laboratory. The laboratory must be equipped to carry out the necessary analyses and approved by the competent authority.
B.
Hygiene requirements
4.
Only the following animal by-products may be transformed in a biogas or composting plant:
(a)
Category 2 material, when using processing method 1 in a Category 2 processing plant;
(b)
[manure and digestive tract content separated from the digestive tract, milk and colostrum, and]
[However, resulting materials from the processing of Category 1 material may be transformed in a biogas plant, provided that the processing was done pursuant to an alternative method approved in accordance with Article 4(2)(e) and, except as otherwise specified, the biogas production is part of that alternative method and the resulting material is disposed of in accordance with the conditions laid down for the alternative method.]
5.
Animal by-products referred to in paragraph 4 must be transformed as soon as possible after arrival. They must be stored properly until treated.
6.
Containers, receptacles and vehicles used for transporting untreated material must be cleaned in a designated area. This area must be situated or designed to prevent risk of contamination of treated products.
7.
Preventive measures against birds, rodents, insects or other vermin must be taken systematically. A documented pest-control programme must be used for that purpose.
8.
Cleaning procedures must be documented and established for all parts of the premises. Suitable equipment and cleaning agents must be provided for cleaning.
9.
Hygiene control must include regular inspections of the environment and equipment. Inspection schedules and results must be documented.
10.
Installations and equipment must be kept in a good state of repair and measuring equipment must be calibrated at regular intervals.
11.
[Digestion residues and compost must be handled and stored at the biogas respective composting plant in such way as to prevent recontamination.]
C.
Processing standards
12.
[Category 3 material used as raw material in a biogas plant equipped with a pasteurisation/hygienisation unit must be submitted to the following minimum requirements:
(a)
maximum particle size before entering the unit: 12 mm;
(b)
minimum temperature in all material in the unit: 70 °C; and
(c)
minimum time in the unit without interruption: 60 minutes.
However, category 3 milk, colostrums and milk products may be used without pasteurisation/hygienisation as raw material in a biogas plant, if the competent authority does not consider them to present a risk of spreading any serious transmissible disease.]
13.
[Category 3 material used as raw material in a composting plant must be submitted to the following minimum requirements:]
(a)
[maximum particle size before entering the composting reactor: 12 mm;]
(b)
[minimum temperature in all material in the reactor: 70 °C; and
(c)
minimum time in the reactor at 70 °C (all material): 60 minutes.]
13a.
[However, the competent authority may authorise the use of other standardised process parameters provided an applicant demonstrates that such parameters ensure minimising of biological risks. That demonstration shall include a validation, which shall be carried out in accordance with points (a) to (f):
(a)
Identification and analysis of possible hazards, including the impact of input material, based on a full definition of the processing conditions.
(b)
A risk assessment, which evaluates how the specific processing conditions referred to in (a) are achieved in practice under normal and atypical situations.
(c)
Validation of the intended process by measuring the reduction of viability/infectivity of:
(i)
endogenous indicator organisms during the process, where the indicator is:
consistently present in the raw material in high numbers,
not less heat resistant to the lethal aspects of the treatment process, but also not significantly more resistant than the pathogens for which it is being used to monitor,
relatively easy to quantify and relatively easy to identify and to confirm;
or
(ii)
a well-characterised test organism or virus, during exposure, introduced in a suitable test body into the starting material.
(d)
The validation of the intended process referred to in (c) must demonstrate that the process achieves the following overall risk reduction:
(i)
for thermal and chemical processes by:
reduction of 5 log10 of Enterococcus faecalis or Salmonella Senftenberg (775W, H2S negative),
reduction of infectivity titre of thermo resistant viruses such as parvovirus by at least 3 log10, whenever they are identified as a relevant hazard;
and
(ii)
as regards chemical processes also by:
(e)
Designing a complete control programme including procedures for monitoring the functioning of the process referred to in (c).
(f)
Measures ensuring continuous monitoring and supervision of the relevant process parameters fixed in the control programme when operating the plant.
Details on the relevant process parameters used in a biogas or composting plant as well as other critical control points must be recorded and maintained so that the owner, operator or their representative and the competent authority can monitor the operation of the plant. Records must be made available to the competent authority on request.
Information relating to a process authorised under this point must be made available to the Commission on request.]
14.
[However, pending the adoption of rules in accordance with Article 6(2)(g), the competent authority may, when catering waste is the only animal by-product used as raw material in a biogas or composting plant, authorise the use of specific requirements other than those laid down in this Chapter provided that they guarantee an equivalent effect regarding the reduction of pathogens. Those specific requirements may also apply to catering waste when it is mixed with manure, digestive tract content separated from the digestive tract, milk and colostrum provided that the resulting material is considered as if it were from catering waste.
Where manure, digestive tract content separated from the digestive tract, milk and colostrum are the only material of animal origin being treated in a biogas or composting plant, the competent authority may authorise the use of specific requirements other than those specified in this Chapter provided that it:
(a)
does not consider that those material present a risk of spreading any serious transmissible disease;
(b)
[considers that the residues or compost are unprocessed material.] ]
D.
Digestion residues and compost
15.
[Representative samples of the digestion residues or compost taken during or immediately after processing at the biogas or composting plant in order to monitor the process must comply with the following standards:
Escherichia coli : n = 5, c = 1, m = 1 000 , M = 5 000 in 1 g;
or
Enterococaceae : n = 5, c = 1, m = 1 000 , M = 5 000 in 1 g;
and
Representative samples of the digestion residues or compost taken during or on withdrawal from storage at the biogas or composting plant must comply with the following standards:
Salmonella : absence in 25 g: n = 5; c = 0; m = 0; M = 0
where:
n
=
number of samples to be tested;
m
=
threshold value for the number of bacteria; the result is considered satisfactory if the number of bacteria in all samples does not exceed m;
M
=
maximum value for the number of bacteria; the result is considered unsatisfactory if the number of bacteria in one or more samples is M or more; and
c
=
number of samples the bacterial count of which may be between m and M, the sample still being considered acceptable if the bacterial count of the other samples is m or less.
Digestion residues or compost, which does not comply with the requirements set out in this Chapter shall be reprocessed, in the case of Salmonella handled or disposed of in accordance with the instructions of the competent authority.]
CHAPTER IIIU.K.Treatment standards for the further processing of rendered fats
The following processes may be used to produce fat derivatives from rendered fats derived from Category 2 material:
1.
transesterification or hydrolysis at least 200 oC, under corresponding appropriate pressure, for 20 minutes (glycerol, fatty acids and esters); or
2.
saponification with NaOH 12M (glycerol and soap):
(a)
in a batch process at 95 oC for three hours; or
(b)
[in a continuous process at 140 °C 2 bars ( 2 000 hPa) for eight minutes, or under equivalent conditions laid down by the Commission. Those measures, designed to amend non-essential elements of this Regulation by supplementing it, shall be adopted in accordance with the regulatory procedure with scrutiny referred to in Article 33(3).]
[However, other processes may be used for further processing of animal fats derived from Category 1 material, provided these processes are approved as alternative method in accordance with Article 4(2)(e).]
ANNEX VIIU.K.SPECIFIC HYGIENE REQUIREMENTS FOR THE PROCESSING AND PLACING ON THE MARKET OF PROCESSED ANIMAL PROTEIN AND OTHER PROCESSED PRODUCTS THAT COULD BE USED AS FEED MATERIAL
CHAPTER IU.K.Specific requirements for the approval of Category 3 processing plants
The following requirements apply in addition to the general requirements laid down in Annex V.
A.
Premises
1.
Premises for the processing of Category 3 material must not be at the same site as premises processing Category 1 or Category 2 material, unless in a completely separate building.
2.
However, the competent authority may authorise the temporary use of a Category 3 processing plant for the processing of Category 1 or Category 2 material when a widespread outbreak of an epizootic disease or other extraordinary and unforeseeable circumstances lead to a lack of capacity at a Category 1 or Category 2 processing plant.
The competent authority must re-approve the Category 3 processing plant in accordance with Article 17 before it processes Category 3 material again.
3.
Category 3 processing plants must have:
(a)
an installation to check the presence of extraneous matter, such as packaging material, metallic pieces, etc. in the animal by-products; and
(b)
if the volume of products treated requires regular or permanent presence of the competent authority, an adequately equipped lockable room for the exclusive use of the inspection service.
B.
Raw material
4.
[Only Category 3 material listed in points (a) to (j) of Article 6(1) that has been handled, stored and transported in accordance with Articles 7, 8 and 9 may be used for the production of processed animal proteins and other feed material.]
5.
Before processing, animal by-products must be checked for the presence of extraneous matter. When present, it must be removed immediately.
C.
Processing standards
6.
The critical control points that determine the extent of the heat treatments applied in processing must be identified for each processing method as specified in Annex V, Chapter III. The critical control points must at least include:
raw material particle size,
temperature achieved in the heat treatment process,
pressure applied to the raw material, if applicable, and
duration of the heat treatment process or feed rate to a continuous system.
Minimum process standards must be specified for each applicable critical control point.
7.
Records must be maintained for at least two years to show that the minimum process values for each critical control point are applied.
8.
Accurately calibrated gauges/recorders must be used to monitor continuously the processing conditions. Records must be kept for at least two years to show the date of calibration of gauges/recorders.
9.
Material that may not have received the specified heat treatment (for example, material discharged at start up, or leakage from cookers) must be recirculated through the heat treatment or collected and reprocessed.
D.
Processed products
10.
Samples of the final products taken during or on withdrawal from storage at the processing plant must comply with the following standards:
Salmonella: absence in 25 g: n = 5, c = 0, m = 0, M = 0
Enterobacteriaceae: n = 5, c = 2, m = 10, M = 300 in 1 g
where:
n
=
number of samples to be tested;
m
=
threshold value for the number of bacteria; the result is considered satisfactory if the number of bacteria in all samples does not exceed m;
M
=
maximum value for the number of bacteria; the result is considered unsatisfactory if the number of bacteria in one or more samples is M or more; and
c
=
number of samples the bacterial count of which may be between m and M, the sample still being considered acceptable if the bacterial count of the other samples is m or less.
11.
[Unused or surplus processed products may after they have been permanently marked:
(a)
be disposed of as waste by incineration or co-incineration in an incineration or co-incineration plant approved in accordance with Article 12;
(b)
be disposed of in a landfill approved under Directive 1999/31/EC; or
(c)
be transformed in a biogas plant or in a composting plant approved in accordance with Article 15.]
CHAPTER IIU.K.Specific requirements for processed animal protein
The following conditions apply in addition to the general conditions laid down in Chapter I.
A.
Processing standards
1.
[ [Mammalian processed animal protein must have been submitted to processing method 1. However, porcine blood or fractions of porcine blood may be submitted instead to any of processing methods 1 to 5 or processing method 7 provided that in the case of processing method 7, a heat treatment throughout its substance at a minimum temperature of 80 °C has been applied.]
However, while the feed ban provided for in Council Decision 2000/766/EC remains in force, mammalian processed animal protein may have been submitted to any of the processing Methods 1 to 5 or Method 7, and shall be permanently marked with a stain or otherwise immediately after that processing, before its disposal as waste in accordance with applicable Community legislation.
In addition, while the feed ban provided for in Council Decision 2000/766/EC remains in force, processed animal protein of mammalian origin exclusively destined for use in petfood, which is transported in dedicated containers that are not used for the transport of animal by-products or feedingstuffs for farmed animals, and which is consigned directly from Category 3 processing plant to the petfood plants, may have been submitted to any of the processing Methods 1 to 5 or 7.]
2.
Non-mammalian processed animal protein, with the exclusion of fishmeal, must have been submitted to any of processing methods 1 to 5 or 7.
3.
Fishmeal must have been submitted:
(a)
to any of the processing methods; or
(b)
to a method and parameters which ensure that the product complies with the microbiological standards set in Chapter I, paragraph 10.
B.
Storage
4.
Processed animal protein must be packed and stored in new or sterilised bags or stored in properly constructed bulk bins.
5.
Sufficient measures must be taken to minimise condensation inside bins, conveyors or elevators.
6.
Products in conveyors, elevators and bins must be protected from casual contamination.
7.
Processed animal protein handling equipment must be maintained in a clean and dry condition and should have adequate inspection points so that equipment can be examined for cleanliness. All storage facilities must be emptied and cleaned regularly, as production requirements require.
8.
Processed animal protein must be kept dry. Leakages and condensation in the storage area must be prevented.
C.
Importation
9.
Member States must authorise the importation of processed animal protein:
(a)
if it comes from third countries that appear on the list in Part II of Annex XI or, in the case of fishmeal, that appear on the list in Part III of Annex XI;
(b)
if it comes from a processing plant that appears on the list referred to in Article 29(4);
(c)
if it has been produced in accordance with this Regulation; and
(d)
[if it is accompanied by a health certificate that conforms to the model set out in Chapter 1 of Annex X.]
10.
Before consignments are released for free circulation within the Community, the competent authority must sample imports of processed animal protein at the border inspection post to ensure compliance with the requirements of Chapter I, paragraph 10. The competent authority must:
(a)
sample each consignment of products carried in bulk; and
(b)
carry out random sampling of consignments of products packaged in the manufacturing plant of origin.
11.
However, when six consecutive tests on bulk consignments originating in a given third country prove negative, the competent authority may carry out random sampling of subsequent bulk consignments from that third country. If one of these random samples proves positive, the competent authority carrying out the sampling must inform the competent authority of the country of origin so that it can take appropriate measures to remedy the situation. The competent authority of the country of origin must bring these measures to the attention of the competent authority carrying out the sampling. In the event of a further positive result from the same source, the competent authority must sample each consignment from the same source until six consecutive tests again prove negative.
12.
Competent authorities must keep a record for at least two years of the results of sampling carried out on all consignments that have undergone sampling.
13.
Where a consignment proves to be positive for salmonella, it must either:
(a)
be dealt with in accordance with the procedure laid down by Article 17(2)(a) of Directive 97/78/EC(); or
(b)
[reprocessed in a processing plant approved pursuant to this Regulation or decontaminated by a treatment authorised by the competent authority. A list of permitted treatments may be established by the Commission. Those measures, designed to amend non-essential elements of this Regulation by supplementing it, shall be adopted in accordance with the regulatory procedure with scrutiny referred to in Article 33(3). The consignment must not be released until it has been treated, tested for salmonella by the competent authority in accordance with Chapter I, paragraph 10, and a negative result obtained.]
CHAPTER IIIU.K.Specific requirements for blood products
The following conditions apply in addition to the general conditions laid down in Chapter I.
A.
Raw material
1.
Only blood coming under paragraph 1(a) and (b) of Article 6 may be used for the production of blood products.
B.
Processing standards
2.
Blood products must have been submitted:
(a)
to any of processing methods 1 to 5 or 7; or
(b)
to a method and parameters which ensure that the product complies with the microbiological standards set in Chapter I, paragraph 10.
C.
Importation
3.
Member States must authorise the importation of blood products if they:
(a)
[come from third countries that appear on the list of part V and part VI of Annex XI as appropriate;]
(b)
come from a processing plant that appears on the list referred to in Article 29(4);
(c)
have been produced in accordance with this Regulation; and
(d)
[if it is accompanied by a health certificate that conforms to the model set out in Chapter 4(B) of Annex X.]
CHAPTER IVU.K.Specific requirements for rendered fats and fish oil
The following conditions apply in addition to the general conditions laid down in Chapter I.
A.
Processing standards
1.
[Unless the rendered fats have been produced in accordance with Chapter II of Annex C to Council Directive 77/99/EEC () , or Chapter 9 of Annex I to Council Directive 92/118/EEC () , rendered fats must be produced using Methods 1 to 5 or Method 7, and fish oils may be produced using Method 6, as referred to in Annex V, Chapter III.
Rendered fats derived from ruminant animals must be purified in such a way that the maximum levels of remaining total insoluble impurities does not exceed 0,15 % in weight.]
B.
Importation of rendered fats
2.
Member States must authorise the importation of rendered fats if they:
(a)
come from third countries appearing on the list in Part IV of Annex XI;
(b)
come from a processing plant that appears on the list referred to in Article 29(4);
(c)
have been produced in accordance with this Regulation;
(d)
either:
(i)
are entirely or partly derived from swine raw material and come from a country or a part of the territory of a country free from foot-and-mouth disease for the previous 24 months and free from classical swine fever and African swine fever for the previous 12 months,
(ii)
are entirely or partly derived from poultry raw material and come from a country or a part of the territory of a country free from Newcastle disease and avian influenza for the previous six months,
(iii)
are entirely or partly derived from ruminant raw material and come from a country or a part of the territory of a country free from foot-and-mouth disease for the previous 24 months and free from Rinderpest for the previous 12 months, or
(iv)
where there has been an outbreak of one of the abovementioned diseases during the relevant period mentioned above, have been subjected to one of the following heat treatment processes:
at least 70 oC for at least 30 minutes, or
at least 90 oC for at least 15 minutes,
and details of the critical control points are recorded and maintained so that the owner, operator or their representative and, as necessary, the competent authority can monitor the operation of the plant. The information must include the particle size, critical temperature and, as appropriate, the absolute time, pressure profile, raw material feed-rate and fat recycling rate; and
(e)
[are accompanied by a health certificate that conforms to the model set out in Chapter 10(A) of Annex X.]
C.
Importation of fish oil
3.
Member States must authorise the importation of fish oil if it:
(a)
comes from third countries appearing on the list in Part III of Annex XI;
(b)
comes from a processing plant that appears on the list referred to in Article 29(4);
(c)
has been produced in accordance with this Regulation; and
(d)
[is accompanied by a health certificate that conforms to the model set out in Chapter 9 of Annex X.]
D.
Hygiene requirements
4.
Where rendered fat or fish oil is packaged, it must be packaged in new containers or in containers that have been cleaned, and all precautions must be taken to prevent its recontamination. Where bulk transport of the products is intended, the pipe, pumps and bulk tanks and any other bulk container or bulk road tanker used in the transportation of the products from the manufacturing plant either directly on to the ship or into shore tanks or direct to plants must have been inspected and found to be clean before use.
[ [CHAPTER V U.K. Specific requirements for milk, milk products, colostrum and colostrum products]
The following conditions apply in addition to the general conditions laid down in Chapter I.
A. Processing standards U.K.
1. Milk must be subjected to one of the following treatments: U.K.
1.1.
sterilisation at an F 0 () value of three or more;
1.2.
UHT () combined with one of the following:
(a)
a subsequent physical treatment, by:
(i)
a drying process, combined in the case of milk intended for feeding with additional heating to 72 °C or more; or
(ii)
lowering the pH below 6 for at least 1 hour;
(b)
the condition that the milk or milk product has been produced at least 21 days before shipping and that during that period no case of foot-and-mouth disease has been detected in the Member State of origin;
1.3.
HTST () applied twice;
1.4.
HTST () in combination with one of the following:
(a)
a subsequent physical treatment, by:
(i)
a drying process, combined in the case of milk intended for feeding with additional heating to 72 °C or more; or
(ii)
lowering the pH below 6 for at least 1 hour;
(b)
the condition that the milk or milk product has been produced at least 21 days before shipping and that during that period no case of foot-and-mouth disease has been detected in the Member State of origin.
2. Milk products must either be subjected to at least one of the treatments provided for in paragraph 1 or be produced from milk treated in accordance with paragraph 1. U.K.
[3. Whey to be fed to animals of species susceptible to foot-and-mouth disease and produced from milk treated in accordance with paragraph 1 must: U.K.
(a)
either be collected at least 16 hours following milk clotting and its pH must be recorded as below 6,0 before transport to animal holdings; or
(b)
have been produced at least 21 days before shipping and during that period no case of foot-and-mouth disease has been detected in the Member State of origin.]
4. In addition to the requirements laid down in paragraphs 1, 2 and 3, milk and milk products must meet the following requirements: U.K.
4.1.
after completion of the processing, every precaution must be taken to prevent contamination of the products;
4.2.
the final product must be labelled so as to indicate that it contains Category 3 material and is not intended for human consumption, and
(a)
packed in new containers; or
(b)
transported in bulk in containers or other means of transport that before use were thoroughly cleansed and disinfected using a disinfectant approved for the purpose by the competent authority.
[5. Raw milk and colostrum must be produced under conditions offering adequate guarantees as regards animal health. Such conditions may be established by the Commission. Those measures, designed to amend non-essential elements of this Regulation by supplementing it, shall be adopted in accordance with the regulatory procedure with scrutiny referred to in Article 33(3).] U.K.
[6. Colostrum and colostrum products must: U.K.
6.1.
be obtained from bovine animals kept on a holding on which all bovine herds are recognised as officially tuberculosis-free, officially brucellosis-free and officially enzootic-bovine-leukosis-free as defined in Article 2(2)(d), (f) and (j) of Directive 64/432/EEC;
6.2.
have been produced at least 21 days before shipping and during that period no case of foot-and-mouth disease has been detected in the Member State of origin;
6.3.
have undergone a single HTST treatment () ;
6.4.
comply with the requirements set out in paragraph 4.]
B. Importation U.K.
1. Member States shall authorise imports of milk and milk products subject to compliance with the following conditions: U.K.
1.1.
[they come from third countries appearing on the list in Part I(A) of Annex XI;]
1.2.
they come from a processing plant which appears on the list referred to in Article 29(4);
1.3.
they are accompanied by a health certificate conforming to the model laid down in Chapter 2 of Annex X;
1.4.
they have undergone at least one of the treatments provided for in paragraphs 1.1, 1.2, 1.3 and point (a) of paragraph 1.4 of Part A;
1.5.
they comply with paragraphs 2 and 4, and, in the case of whey, paragraph 3 of Part A.
[2. By way of derogation from paragraph 1.4, Member States shall authorise imports of milk and milk products from third countries so authorised in Column ‘ A ’ of Annex I to Commission Decision 2004/438/EC () provided that the milk and milk products have undergone a single HTST treatment and: U.K.
(a)
have not been shipped before a period of at least 21 days has elapsed after production and during that period no case of foot-and-mouth disease has been detected in the exporting third country; or
(b)
have been presented at an EU border inspection post at least 21 days after production and during that period no case of foot-and-mouth disease has been detected in the exporting third country.]
[2a. Member States shall authorise imports of colostrum or colostrum products of bovine animals provided that: U.K.
they come from a third country appearing on the list in Part I(B) of Annex XI;
they comply with the conditions set out in paragraphs 1.2 and 1.3;
they have undergone a single HTST treatment () and:
(a)
have not been shipped before a period of at least 21 days has elapsed after production and during that period no case of foot-and-mouth disease has been detected in the exporting third country; or
(b)
have been presented at an EU border inspection post at least 21 days after production and during that period no case of foot-and-mouth disease has been detected in the exporting third country;
they have been obtained from bovine animals subject to regular veterinary inspections to ensure that animals come from holdings on which all bovine herds are:
(a)
either recognised as officially tuberculosis free and officially brucellosis free as defined in Article 2(2)(d) and (f) of Directive 64/432/EEC or not restricted under the national legislation of the third country of origin of the colostrum regarding eradication of tuberculosis and brucellosis; and
(b)
either recognised as official enzootic-bovine-leukosis-free as defined in Article 2(2)(j) of Directive 64/432/EEC or included in an official system for the control of enzootic-bovine-leukosis and there has been no evidence as a result of clinical and laboratory testing of this disease in the herd during the past two years;
after completion of the processing, every precaution has been taken to prevent contamination of the colostrum or colostrum products;
the final product has been labelled so as to indicate that it contains Category 3 material and is not intended for human consumption, and that it has been:
(a)
packed in new containers; or
(b)
transported in bulk in containers or other means of transport that before use were thoroughly cleansed and disinfected using a disinfectant approved for the purpose by the competent authority.]
[3. Where a risk of introduction of an exotic disease or any other risk to animal health is identified, additional conditions for the protection of animal health may be established by the Commission. Those measures, designed to amend non-essential elements of this Regulation by supplementing it, shall be adopted in accordance with the regulatory procedure with scrutiny referred to in Article 33(3).] ] U.K.
CHAPTER VIU.K.Specific requirements for gelatin and hydrolysed protein
The following conditions apply in addition to the general conditions laid down in Chapter I.
A.
Processing standards for gelatin
1.
(a)
Gelatin must be produced by a process that ensures that unprocessed Category 3 material is subjected to a treatment with acid or alkali, followed by one or more rinses. The pH must be adjusted subsequently. Gelatin must be extracted by heating one or several times in succession, followed by purification by means of filtration and sterilisation.
(b)
After having been subjected to the processes referred to in subparagraph (a), gelatin may undergo a drying process and, where appropriate, a process of pulverisation or lamination.
(c)
The use of preservatives, other than sulphur dioxide and hydrogen peroxide, is prohibited.
2.
Gelatin must be wrapped, packaged, stored and transported under satisfactory hygiene conditions. In particular:
(a)
a room must be provided for storing materials for wrapping and packaging;
(b)
wrapping and packaging must take place in a room or in a place intended for that purpose;
and
(c)
wrappings and packages containing gelatin must carry the words ‘gelatin suitable for animal consumption’.
B.
Processing standards for hydrolysed protein
3.
[Hydrolysed protein must be produced using a production process involving appropriate measures to minimise contamination of raw Category 3 material. Hydrolysed protein shall have a molecular weight below 10 000 Dalton.
In addition, hydrolysed proteins entirely or partly derived from ruminants hides and skins shall be produced in a processing plant dedicated only to hydrolysed protein production, using a process involving the preparation of raw Category 3 material by brining, liming and intensive washing followed by:
(a)
exposure of the material to a pH of more than 11 for more than three hours at a temperature of more than 80 o C and subsequently by heat treatment at more than 140 o C for 30 minutes at more than 3,6 bar;
(b)
exposure of the material to a pH of 1 to 2, followed by a pH of more than 11, followed by heat treatment at 140 o C for 30 minutes at 3 bar; or
(c)
[an equivalent production process approved by the Commission. Those measures, designed to amend non-essential elements of this Regulation by supplementing it, shall be adopted in accordance with the regulatory procedure with scrutiny referred to in Article 33(3).] ]
C.
Importation
4.
[Member States must authorise the importation of gelatine and hydrolysed proteins if they:
(a)
come from third countries that appear on the list in Part XI of Annex XI;
(b)
come from a processing plant that appears on the list referred to in Article 29(4);
(c)
have been produced in accordance with this Regulation; and
(d)
[are accompanied by a health certificate that conforms to the models set out in Chapter 11 and Chapter 12 of Annex X as appropriate.] ]
[CHAPTER VII U.K. Specific requirements for dicalcium phosphate
The following conditions apply in addition to the general conditions laid down in Chapter I.
A. Processing standards U.K.
[1. Dicalcium phosphate must be produced by a process that: U.K.
(a)
ensures that all Category 3 bone-material is finely crushed and degreased with hot water and treated with dilute hydrochloric acid (at a minimum concentration of 4 % and a pH of less than 1,5) over a period of at least two days;
(b)
following the procedure provided for in point (a), applies a treatment of the obtained phosphoric liquor with lime, resulting in a precipitate of dicalcium phosphate at pH 4 to 7; and
(c)
finally, air-dries the precipitate of dicalcium phosphate with inlet temperature of 65 °C to 325 °C and end temperature between 30 °C and 65 °C, or
by an equivalent process approved by the Commission. Those measures, designed to amend non-essential elements of this Regulation by supplementing it, shall be adopted in accordance with the regulatory procedure with scrutiny referred to in Article 33(3).]
2. Where dicalcium phosphate is derived from defatted bones it shall be derived from bones fit for human consumption following ante and post-mortem inspection. U.K.
B. Importation U.K.
3. Member States must authorise the importation of dicalcium phosphate if it: U.K.
(a)
comes from third countries that appear on the list in Part XI of Annex XI;
(b)
comes from a processing plant that appears on the list referred to in Article 29(4);
(c)
has been produced in accordance with this Regulation; and
(d)
[is accompanied by a health certificate that conforms to the model set out in Chapter 12 of Annex X.] ]
[CHAPTER VIII U.K. Specific requirements for tricalcium phosphate
The following conditions apply in addition to the general conditions laid down in Chapter I.
A. Processing standards U.K.
[1. Tricalcium phosphate must be produced by a process that ensures: U.K.
(a)
that all Category 3 bone-material is finely crushed and degreased in counter-flow with hot water (bone chips less than 14 mm);
(b)
continuous cooking with steam at 145 °C during 30 minutes at 4 bars;
(c)
separation of the protein broth from the hydroxyapatite (tricalcium phosphate) by centrifugation; and
(d)
granulation of the tricalcium phosphate after drying in a fluid bed with air at 200 °C; or
by an equivalent production process approved by the Commission. Those measures, designed to amend non-essential elements of this Regulation by supplementing it, shall be adopted in accordance with the regulatory procedure with scrutiny referred to in Article 33(3).]
B. Importation U.K.
2. Member States must authorise the importation of tricalcium phosphate if it: U.K.
(a)
comes from third countries that appear on the list in Part XI of Annex XI;
(b)
comes from a processing plant that appears on the list referred to in Article 29(4);
(c)
has been produced in accordance with this Regulation; and
(d)
[is accompanied by a health certificate that conforms to the model set out in Chapter 12 of Annex X.] ]
[CHAPTER IX U.K. Specific requirements for collagen
The following conditions apply in addition to the general conditions laid down in Chapter I.
A. Processing standards U.K.
1. Collagen must be produced by a process ensuring that unprocessed category 3 material is subjected to a treatment involving washing, pH adjustment using acid or alkali followed by one or more rinses, filtration and extrusion. After that treatment collagen may undergo a drying process. U.K.
2. The use of preservatives, other than those permitted under Community legislation shall be prohibited. U.K.
3. Collagen must be wrapped, packaged, stored and transported under satisfactory hygiene conditions. In particular: U.K.
(a)
a room must be provided for storing materials for wrapping and packaging;
(b)
wrapping and packaging must take place in a room or in a place intended for that purpose; and
(c)
wrapping and packages containing collagen must be labelled with the words ‘ collagen suitable for animal consumption ’ .
B. Importation U.K.
4. Member States must authorise the importation of collagen if it: U.K.
(a)
comes from a third country that appears on a Community list set out in Part XI of Annex XI;
(b)
comes from a plant that appears on the list referred to in Article 29(4);
(c)
has been produced in accordance with this Regulation; and
(d)
is accompanied by a health certificate that conforms to the model set out in Chapter 11 of Annex X.
CHAPTER X U.K. Specific requirements for egg products
The following conditions apply in addition to the general conditions laid down in Chapter I.
A. Processing standards U.K.
1. Egg products must have been: U.K.
(a)
submitted to any of processing Methods 1 to 5 or 7; or
(b)
submitted to a method and parameters which ensure that the products comply with the microbiological standards set in Chapter I, paragraph 10; or
(c)
treated in accordance with Chapter V of the Annex to Council Directive 89/437/EC () laying down hygiene and health problems affecting the production and the placing on the market of egg products.
B. Importation U.K.
2. Member States must authorise the importation of egg products if they: U.K.
(a)
come from a third country that appears on a Community list set out in Part XVI of Annex XI;
(b)
come from a plant that appears on the list referred to in Article 29(4);
(c)
have been produced in accordance with this Regulation; and
(d)
are accompanied by a health certificate that conforms to the model set out in Chapter 15 of Annex X.]
ANNEX VIIIU.K.REQUIREMENTS FOR THE PLACING ON THE MARKET OF PETFOOD, DOGCHEWS AND TECHNICAL PRODUCTS
CHAPTER IU.K.Requirements for the approval of petfood and technical plants
Plants producing petfood, dogchews and technical products, other than organic fertilizers, soil improvers and fat derivatives derived from Category 2 material, must fulfil the following requirements:
1.
they must have adequate facilities for storing and treating incoming material in complete safety; and
2.
they must have adequate facilities for disposing of unused animal by-products remaining after the production of the products in accordance with this Regulation, or this material must be sent to a processing plant or to an incineration or co-incineration plant in accordance with this Regulation.
CHAPTER IIU.K.Requirements for petfood and dogchews
A.Raw materialU.K.
[1. The only animal by-products that may be used to produce petfood and dogchews are those referred to in Article 6(1)(a) to (j). However, raw petfood may only be manufactured from animal by-products referred to in Article 6(1)(a) or Article 6(1)(b).] U.K.
B.Processing standardsU.K.
2.Canned petfood must be subjected to heat treatment to a minimum Fc value of 3.U.K.
[3. Processed petfood other than canned petfood must: U.K.
(a)
be subjected to a heat treatment of at least 90 °C throughout the substance of the final product;
(b)
be subjected to a heat treatment to at least 90 °C of the ingredients of animal origin; or
(c)
be produced as regards ingredients of animal origin exclusively using:
(i)
meat or meat products which have been subject to a heat treatment of at least 90 °C throughout their substance;
(ii)
the following animal by-products or processed products which have been processed in accordance with the requirements of this Regulation: milk and milk based products, gelatine, hydrolysed protein, egg products, collagen, blood products, processed animal protein including fishmeal, rendered fat, fish oils, dicalcium phosphate, tricalcium phosphate or flavouring innards.
After the heat treatment, every precaution must be taken to ensure that such processed petfood is not exposed to contamination.
The processed petfood must be packaged in new packaging.]
[4. Dogchews must be subjected to a treatment during processing sufficient to destroy pathogenic organisms, including salmonella. After that treatment, every precaution must be taken to ensure that such dogchews are not exposed to contamination. The dogchews must be packed in new packaging.] U.K.
5.Raw petfood must be packed in new packaging preventing any leakage. Effective steps must be taken to ensure that the product is not exposed to contamination throughout the production chain and up to the point of sale. The wording ‘petfood only’ must be visibly and legibly displayed on the packaging.U.K.
[6. Random samples must be taken during production and/or during storage (before dispatch) to verify compliance with the following standards: U.K.
Salmonella : absence in 25 g, n = 5, c = 0, m = 0, M = 0.
Enterobacteriaceae : n = 5, c = 2, m = 10, M = 300 in 1 g
Where:
n
=
number of samples to be tested;
m
=
threshold value for the number of bacteria; the result is considered satisfactory if the number of bacteria in all samples does not exceed m;
M
=
maximum value for the number of bacteria; the result is considered unsatisfactory if the number of bacteria in one or more samples is M or more; and
c
=
number of samples the bacterial count of which may be between m and M, the sample still being considered acceptable if the bacterial count of the other samples is m or less.
However, for canned petfood that has undergone the heat treatment referred to in paragraph 2, sampling and testing for Salmonella and Enterobacteriaceae may not be necessary.]
C.ImportationU.K.
7.Member States must authorise importation of petfood and dogchews if they:U.K.
(a)
come from third countries that appear on the list in Part X of Annex XI;
(b)
come from petfood plants approved by the competent authority of the third country meeting the specific conditions laid down in this Regulation;
(c)
have been produced in accordance with this Regulation;
(d)
are accompanied:
(i)
in the case of canned petfood, by a certificate that conforms to the model laid down in Chapter 3(A) of Annex X,
(ii)
in the case of processed petfood other than canned petfood, by a certificate that conforms to the model laid down in Chapter 3(B) of Annex X,
(iii)
in the case of dogchews, by a certificate that conforms to the model laid down in Chapter 3(C) of Annex X, or
(iv)
in the case of raw petfood, by a certificate that conforms to the model laid down in Chapter 3(D) of Annex X.
CHAPTER IIIU.K.Requirements for manure, processed manure and processed manure products
I.Unprocessed manureU.K.
A.TradeU.K.
1.
(a)
Trade in unprocessed manure of species other than poultry or equidae is prohibited, except for manure:
(i)
from an area which is not subject to restrictions by virtue of a serious transmissible disease, and
(ii)
intended for application, under the supervision of the competent authorities, to land forming part of a single holding located on both sides of the border of two Member States.
(b)
However, the competent authority may grant specific approval for the introduction on to its territory of:
(i)
manure intended for processing in a technical plant or a biogas plant or in a composting plant approved by the competent authority in accordance with this Regulation with a view to the manufacture of the products referred to under Section II below. The competent authority must take account of the origin of the manure when approving such plants; or
(ii)
manure intended for applying to land on a holding. Such trade can only occur with the consent of the competent authorities of both the Member States of origin and destination. When considering giving consent, the competent authorities must have particular regard to the origin of the manure, its destination and animal health and safety considerations.
A health certificate conforming to a model laid down under the procedure referred to in Article 33(2) must accompany the manure in such cases.
2.Trade in unprocessed poultry manure is subject to the following conditions:U.K.
(a)
the manure must originate in an area which is not subject to restrictions by virtue of Newcastle disease or avian influenza;
(b)
in addition, unprocessed manure from poultry flocks vaccinated against Newcastle disease must not be dispatched to a region which has obtained Newcastle disease non-vaccinating status pursuant to Article 15(2) of Directive 90/539/EEC(); and
(c)
a health certificate conforming to a model laid down under the procedure referred to in Article 33(2) must accompany the manure.
[3. Unprocessed manure of equidae which is traded must not originate from a holding subject to animal health restrictions pertaining to glanders, vesicular stomatitis, anthrax or rabies in accordance with Article 4 (5) of Directive 90/426/EEC.] U.K.
B.ImportationU.K.
[4. The importation of unprocessed manure is prohibited.] U.K.
II.Processed manure and processed manure productsU.K.
A.Placing on the marketU.K.
[5. The placing on the market of processed manure and processed manure products shall be subject to the following conditions set out in points (a) to (e): U.K.
(a)
They must come from a technical plant, a biogas plant or a composting plant approved by the competent authority in accordance with this Regulation.
(b)
They must have been subjected to a heat treatment process of at least 70 °C for at least 60 minutes and they must have been subjected to reduction in spore-forming bacteria and toxic formation.]
(c)
[However, the competent authority may authorise the use of other standardised process parameters than those described in (b) provided an applicant demonstrates that such parameters ensure minimising of biological risks. This demonstration shall include a validation, which shall be carried out as follows:
(i)
Identification and analysis of possible hazards including the impact of input material, based on a full definition of the processing conditions, and a risk assessment, which evaluates how the specific processing conditions are achieved in practice under normal and atypical situations.
(ii)
Validation of the intended process
by measuring the reduction of viability/infectivity of endogenous indicator organisms during the process, where the indicator is:
consistently present in the raw material in high numbers,
not less heat resistant to the lethal aspects of the treatment process, but also not significantly more resistant than the pathogens for which it is being used to monitor,
relatively easy to quantify and relatively easy to identify and confirm;
or
by measuring the reduction of viability/infectivity, during exposure, of a well-characterised test organism or virus introduced in a suitable test body into the starting material.
(iii)
The validation referred to in point (ii) must demonstrate that the process achieves the following overall risk reduction:
for thermal and chemical processes by reduction of Enterococcus faecalis by at least 5 log10 and by reduction of infectivity titre of thermo resistant viruses such as parvovirus , where they are identified as a relevant hazard, by at least 3 log10,
for chemical processes also by reduction of resistant parasites such as eggs of ascaris sp. by at least 99,9 % (3 log10) of viable stages.
(iv)
Designing a complete control programme including procedures for monitoring the process.
(v)
Measures ensuring continuous monitoring and supervision of the relevant process parameters fixed in the control programme when operating the plant.
Details on the relevant process parameters used in a plant as well as other critical control points must be recorded and maintained so that the owner, operator or their representative and the competent authority can monitor the operation of the plant. Records must be made available to the competent authority on request.
Information relating to a process authorised under this point must be made available to the Commission on request.]
(d)
[Representative samples of the manure taken during or immediately after processing at the plant in order to monitor the process must comply with the following standards:
Escherichia coli : n = 5, c = 5, m = 0, M = 1 000 in 1 g;
or
Enterococaceae : n = 5, c = 5, m = 0, M = 1 000 in 1 g;
and
Representative samples of the manure taken during or on withdrawal from storage at the technical, biogas or composting plant must comply with the following standards:
Salmonella : absence in 25 g: n = 5; c = 0; m = 0; M = 0
where:
n
=
number of samples to be tested;
m
=
threshold value for the number of bacteria; the result is considered satisfactory if the number of bacteria in all samples does not exceed m;
M
=
maximum value for the number of bacteria; the result is considered unsatisfactory if the number of bacteria in one or more samples is M or more; and
c
=
number of samples the bacterial count of which may be between m and M, the sample still being considered acceptable if the bacterial count of the other samples is m or less.
Processed manure or processed manure products not complying with the above requirements shall be regarded as unprocessed;
(e)
They must be stored in such a way that once processed contamination or secondary infection and dampness is minimised. They must therefore be stored in:
(i)
well-sealed and insulated silos, or
(ii)
properly sealed packs (plastic bags or ‘ big bags ’ ).]
B.ImportationU.K.
6.Member States must authorise importation of processed manure and processed manure products if they:U.K.
(a)
come from third countries that appear on the list in Part IX of Annex XI;
(b)
come from a plant approved by the competent authority of the third country meeting the specific conditions laid down in this Regulation;
(c)
satisfy the requirements of paragraph 5 above; and
(d)
[are accompanied by a health certificate that conforms to the model laid down in Chapter 17 of Annex X.]
III.GuanoU.K.
7.The placing on the market of ‘guano’ is not subject to any animal health conditions.U.K.
[CHAPTER IV U.K. Requirements for blood and blood products, excluding from equidae, for the manufacture of technical products
A. Importation U.K.
1. Imports of blood are subject to the requirements laid down in Chapter XI. U.K.
2. Member States must authorise imports of blood products for the manufacture of technical products, including material originating from animals to which substances prohibited pursuant to Directive 96/22/EC have been administered, if they: U.K.
(a)
come from third countries that appear on the list in Part VI (A) of Annex XI as applicable;
(b)
come from a technical plant meeting the specific conditions laid down in this Regulation or from the establishment of collection;
(c)
are accompanied by a health certificate that conforms to the model set out in Chapter 4(C) or Chapter 4(D) of Annex X as appropriate.
3. The blood from which blood products for the manufacture of technical products are produced must have been collected: U.K.
(a)
in slaughterhouses approved in accordance with Community legislation;
(b)
in slaughterhouses approved and supervised by the competent authority of the third country; or
(c)
from live animals in facilities approved and supervised by the competent authority of the third country.
4. In the case of blood products for the manufacture of technical products which have been derived from animals belonging to the taxa Artiodactyla, Perissodactyla and Proboscidea , including their crossbreeds, they must comply with the conditions of either points (a) or (b): U.K.
(a)
the products have undergone one of the following treatments guaranteeing the absence of pathogens of the diseases referred to in point (b):
(i)
heat treatment at a temperature of 65 °C for at least three hours, followed by an effectiveness check,
(ii)
irradiation at 25 kGy by gamma rays, followed by an effectiveness check,
(iii)
heat treatment of at least 80 °C throughout their substance, followed by an effectiveness check,
(iv)
in the case of animals other than Suidae and Tayassuidae only: change in pH to pH 5 for two hours, followed by an effectiveness check;
(b)
in case of blood products not treated in accordance with point (a) the products originate from a country or region:
(i)
where no case of rinderpest, peste des petits ruminants and Rift Valley fever has been recorded for 12 months and in which vaccination has not been carried out against those diseases for at least 12 months,
(ii)
where no case of foot-and-mouth disease has been recorded for 12 months and in which vaccination has not been carried out against this disease for at least 12 months, or
where no case of foot-and-mouth disease has been recorded for 12 months and in which vaccination programmes against foot-and-mouth disease are being officially carried out and controlled in domestic ruminant animals for at least 12 months; in this case, following the border check provided for in Directive 97/78/EC, and in accordance with the conditions laid down in Article 8(4) of that Directive, the products must be transported directly to the technical plant of destination and all precautions, including safe disposal of waste, unused or surplus material, must be taken to avoid risks of spreading diseases to animals or humans.
In addition to points (i) and (ii), in the case of animals other than Suidae and Tayassuidae , one of the following conditions must be complied with:
in the country or region of origin no case of vesicular stomatitis and bluetongue (including the presence of seropositive animals) has been recorded for 12 months and vaccination has not been carried out against those diseases for at least 12 months in the susceptible species,
following the border check provided for in Directive 97/78/EC, and in accordance with the conditions laid down in Article 8(4) of that Directive, the products must be transported directly to the technical plant of destination and all precautions, including safe disposal of waste, unused or surplus material, must be taken to avoid risks of spreading diseases to animals or humans.
In addition to points (i) and (ii), in the case of Suidae and Tayassuidae , in the country or region of origin no case of swine vesicular disease, classical swine fever and African swine fever has been recorded for at least 12 months, vaccination has not been carried out against those diseases for at least 12 months and one of the following conditions are complied with:
in the country or region of origin no case of vesicular stomatitis (including the presence of seropositive animals) has been recorded for 12 months and vaccination has not been carried out against this disease for at least 12 months in the susceptible species,
following the border check provided for in Directive 97/78/EC, and in accordance with the conditions laid down in Article 8(4) of that Directive, the products must be transported directly to the technical plant of destination and all precautions, including safe disposal of waste, unused or surplus material, must be taken to avoid risks of spreading diseases to animals or humans.
5. In the case of blood products for the manufacture of technical products which have been derived from poultry and other avian species, they must comply with the conditions of either points (a) or (b): U.K.
(a)
the products have undergone one of the following treatments guaranteeing the absence of pathogens of the diseases referred to in point (b):
(i)
heat treatment at a temperature of 65 °C for at least three hours, followed by an effectiveness check,
(ii)
irradiation at 25 kGy by gamma rays, followed by an effectiveness check,
(iii)
heat treatment of at least 70 °C throughout their substance, followed by an effectiveness check;
(b)
in case of blood products not treated in accordance with point (a) the products originate from a country or region:
(i)
which has been free from Newcastle disease and highly pathogenic avian influenza as defined in the Terrestrial Animal Health Code of the OIE,
(ii)
which during the last 12 months has not carried out vaccination against avian influenza,
(iii)
where the poultry or other avian species from which the products derive have not been vaccinated against Newcastle disease with vaccines prepared from a Newcastle disease master strain showing a higher pathogenicity than lentogenic virus strains.]
[CHAPTER V U.K. Requirements for blood and blood products from equidae for technical purposes
A. Placing on the market U.K.
The placing on the market for technical purposes of blood and blood products from equidae shall be subject to the following conditions:
1.
Blood may be placed on the market provided that:
(a)
it has been collected from equidae which:
(i)
at inspection on the date of blood collection do not show clinical signs of any of the compulsorily notifiable diseases listed in Annex A to Directive 90/426/EEC and of Equine influenza, Equine piroplasmosis, Equine rhinopneumonitis and Equine viral arteritis listed in point 4 of Article 1.2.3. of the Terrestrial Animal Health Code of the World Organisation for Animal Health (OIE), 2009 Edition;
(ii)
have been kept for at least 30 days prior to the date of and during blood collection on holdings under veterinary supervision which were not subject to a prohibition order pursuant to Article 4(5) of Directive 90/426/EEC or restrictions pursuant to Article 5 thereof;
(iii)
for the periods laid down in Article 4(5) of Directive 90/426/EEC had no contact with equidae from holdings which were subject to a prohibition order for animal health reasons pursuant to that Article and for at least 40 days prior to the date of and during blood collection had no contact with equidae from a Member State or third country not considered free of African horse sickness in accordance with Article 5(2)(a) of that Directive;
(b)
it has been collected under veterinary supervision either:
(i)
in slaughterhouses approved in accordance with Regulation (EC) No 853/2004; or
(ii)
in facilities approved, furnished with a veterinary approval number and supervised by the competent authority for the purpose of collecting blood from equidae for the production of blood products for technical purposes.
2.
Blood products may be placed on the market provided that:
(a)
all precautions have been taken to avoid contamination of the blood products with pathogenic agents during production, handling and packaging;
(b)
the blood products have been produced from blood which:
(i)
either fulfils the conditions set out in paragraph 1(a); or
(ii)
has been subjected to at least one of the following treatments, followed by an effectiveness check, for the inactivation of possible causative pathogens for African horse sickness, equine encephalomyelitis of all types including Venezuelan equine encephalomyelitis, equine infectious anaemia, vesicular stomatitis and glanders ( Burkholderia mallei ):
heat treatment at a temperature of 65 °C for at least three hours;
irradiation at 25 kGy by gamma rays;
change in pH to pH 5 for two hours;
heat treatment of at least 80 °C throughout their substance.
3.
Blood and blood products from equidae must be packed in sealed impermeable containers which:
(a)
are clearly labelled ‘ BLOOD AND BLOOD PRODUCTS FROM EQUIDAE, NOT FOR HUMAN OR ANIMAL CONSUMPTION ’ ;
(b)
bear the approval number of the establishment of collection referred to in paragraph 1(b).
B. Importation U.K.
Member States shall authorise imports of blood and blood products from equidae for technical purposes subject to the following conditions:
1.
The blood must comply with the conditions set out in paragraph 1(a) of Section A and must be collected under veterinary supervision either in:
(a)
slaughterhouses
(i)
approved in accordance with Regulation (EC) No 853/2004; or
(ii)
approved and supervised by the competent authority of the third country; or
(b)
facilities approved, furnished with a veterinary approval number and supervised by the competent authority of the third country for the purpose of collecting blood from equidae for the production of blood products for technical purposes.
2.
The blood products must comply with the conditions set out in paragraph 2 of Section A.
In addition, the blood products referred to in paragraph 2(b)(i) of Section A must have been produced from blood collected from equidae which have been kept for a period of at least three months, or since birth if less than three months old, prior to the date of collection on holdings under veterinary supervision in the third country of collection which during that period and the period of blood collection has been free of:
(a)
African horse sickness in accordance with Article 5(2)(a) of Directive 90/426/EEC;
(b)
Venezuelan equine encephalomyelitis for a period of at least two years;
(c)
glanders:
(i)
for a period of three years; or
(ii)
for a period of six months where the animals have shown no clinical signs of glanders ( Burkholderia mallei ) during the post-mortem inspection in the slaughterhouse referred to in paragraph 1(a), including a careful examination of mucous membranes from the trachea, larynx, nasal cavities and sinuses and their ramifications, after splitting the head in the median plane and excising the nasal septum;
(d)
vesicular stomatitis for six months.
3.
Blood products must come from a technical plant approved by the competent authority of the third country meeting the specific conditions laid down in Article 18 of Regulation (EC) No 1774/2002.
4.
Blood and blood products must come from a third country that appears on the list referred to in the following Parts of Annex XI:
(a)
Part XIII(A) where blood has been collected in accordance with paragraph 1 of Section A or where blood products have been produced in accordance with paragraph 2(b)(i) of Section A; or
(b)
Part XIII(B) where they have been treated in accordance with paragraph 2(b)(ii) of Section A.
5.
Blood and blood products shall be packed and labelled in accordance with paragraph 3(a) of Section A and shall be accompanied by a health certificate that conforms to the model set out in Chapter 4(A) of Annex X, duly completed and signed by the official veterinarian.]
CHAPTER VIU.K.Requirements for hides and skins of ungulates
A.ScopeU.K.
1.The provisions of this Chapter do not apply:U.K.
(a)
[to hides and skins of ungulates complying with the requirements of Regulation (EC) No 853/2004 of 29 April 2004 of the European Parliament and of the Council laying down specific hygiene rules for food of animal origin () ;]
(b)
to hides and skins of ungulates having undergone the complete process of tanning;
(d)
to ‘pickled pelts’; and
(e)
to limed hides (treated with lime and in brine at a pH of 12 to 13 for at least eight hours).
2.Within the scope defined in paragraph 1, the provisions of this Chapter apply to fresh, chilled and treated hides and skins. For the purpose of this Chapter, ‘treated hides and skins’ means hides and skins that have been:U.K.
(b)
dry-salted or wet-salted for at least 14 days prior to dispatch;
(c)
salted for seven days in sea salt with the addition of 2 % of sodium carbonate;
(d)
dried for 42 days at a temperature of at least 20 oC; or
(e)
[preserved by a process other than tanning specified by the Commission. Those measures, designed to amend non-essential elements of this Regulation, shall be adopted in accordance with the regulatory procedure with scrutiny referred to in Article 33(3).]
B.TradeU.K.
[3. Trade in fresh or chilled hides and skins is subject to the same health conditions as those applicable to fresh meat pursuant to Council Directive 2002/99/EC of 16 December 2002 laying down the animal health rules governing the production, processing, distribution and introduction of products of animal origin for human consumption () .] U.K.
4.Trade in treated hides and skins is authorised on condition that the commercial document provided for in Annex II accompanies each consignment and attests that:U.K.
(a)
the hides and skins have been treated in accordance with paragraph 2; and
(b)
the consignment has not been in contact with other animal products or live animals presenting a risk of spreading a serious transmissible disease.
C.ImportationU.K.
5.Member States must authorise the import of fresh or chilled hides and skins if:U.K.
(a)
they have been obtained from animals referred to in Article 6(1)(b) or (c);
(b)
[ [they come from a third country or, in the case of regionalisation in accordance with Community legislation, from a part of a third country, appearing on the list set out in Part XIV(A) of Annex XI and which, as appropriate to the species concerned:]
(i)
for at least 12 months before dispatch, has been free from the following diseases:
classical swine fever,
African swine fever, and
rinderpest, and
(ii)
has been free for at least 12 months before dispatch from foot-and-mouth disease and where, for 12 months before dispatch, no vaccination has been carried out against foot-and-mouth disease;]
(c)
they have been obtained from:
(i)
animals that have remained in the territory of the country of origin for at least three months before being slaughtered or since birth in the case of animals less that three months old,
(ii)
in the case of hides and skins from bi-ungulates, animals that come from holdings in which there has been no outbreak of foot-and-mouth disease in the previous 30 days, and around which within a radius of 10 km there has been no case of foot-and-mouth disease for 30 days,
(iii)
in the case of hides and skins from swine, animals that come from holdings in which there has been no outbreak of swine vesicular disease in the previous 30 days, or of classical or African swine fever in the previous 40 days, and around which within a radius of 10 km there has been no case of these diseases for 30 days, or
(iv)
animals that have passed the ante-mortem health inspection at the slaughterhouse during the 24 hours before slaughter and have shown no evidence of foot-and-mouth disease, rinderpest, classical swine fever, African swine fever or swine vesicular disease;
(d)
they have undergone all precautions to avoid recontamination with pathogenic agents; and
(e)
a certificate conforming to the model laid down in Chapter 5(A) of Annex X accompanies them.
6.Member States must authorise the import of treated hides and skins if:U.K.
(a)
they have been obtained from animals referred to in Article 6(1)(b), (c) or (k);
(b)
[they come either from:
(i)
a third country or, in the case of regionalisation in accordance with Community legislation, from a part of a third country, appearing on the list set out in Part XIV(B) of Annex XI from which imports of fresh meat of the corresponding species are authorised and they have been treated in accordance with paragraph 2(a), (b) and (c) of A; or
(ii)
a third country appearing on the list set out in Part XIV(B) of Annex XI and they have been treated in accordance with paragraph 2(c) or (d) of A; or
(iii)
equidae or ruminant animals from a third country appearing on the list set out in Part XIV(C) of Annex XI, which have been treated in accordance with paragraph 2(a), (b) and (c) of A and after treatment have been kept separate for at least 21 days;
(c)
in the case of salted hides and skins transported by ship, they have been treated in accordance with paragraphs 2(b) or (c) of A and have been kept separated after treatment during transportation for at least 14 days in the case of paragraph (b) or seven days in the case of paragraph (c) before importation and the health certificate accompanying the consignment attests such treatment and the duration of the transportation; and
(d)
a health certificate conforming to the model health certificate laid down in Chapter 5(B) of Annex X, or, in the case of hides and skins referred to in paragraph 6(b)(iii) of C of this Annex, an official declaration conforming to the model laid down in Chapter 5 (C) of Annex X, accompanies them.]
7.Fresh, chilled or treated hides and skins of ungulates must be imported in containers, road vehicles, railway wagons or bales sealed by the competent authority of the third country of dispatch.U.K.
CHAPTER VIIU.K.Requirements for game trophies
A.Raw materialU.K.
1.Without prejudice to the measures adopted pursuant to Council Regulation (EC) No 338/97 of 9 December 1996 on the protection of species of wild fauna and flora by regulating trade therein(), game trophies:U.K.
(a)
of ungulates and birds having undergone a complete taxidermy treatment ensuring their preservation at ambient temperatures; and
(b)
of species other than ungulates and birds,
are not subject to any ban or restriction for reasons of animal health.
2.Without prejudice to the measures adopted pursuant to Regulation (EC) No 338/97, game trophies of ungulates and birds not having undergone the treatment mentioned in paragraph 1(a) are subject to the following conditions. They must:U.K.
(a)
come from animals originating in an area not subject to restrictions as a result of the presence of serious transmissible diseases to which animals of the species concerned are susceptible; or
(b)
comply with the conditions laid down in paragraphs 3 or 4 if they come from animals originating in an area subject to restrictions as a result of the presence of serious transmissible diseases to which animals of the species concerned are susceptible.
3.In respect of game trophies consisting solely of bone, horns, hooves, claws, antlers or teeth, the trophies must:U.K.
(a)
have been immersed in boiling water for an appropriate time so as to ensure that any matter other than bone, horns, hooves, claws, antlers or teeth is removed;
(b)
have been disinfected with a product authorised by the competent authority, in particular with hydrogen peroxide where parts consisting of bone are concerned;
(c)
be packaged, immediately after treatment, without being in contact with other products of animal origin likely to contaminate them, in individual, transparent and closed packages so as to avoid any subsequent contamination; and
(d)
be accompanied by a document or certificate certifying that the above conditions have been met.
4.In respect of game trophies consisting solely of hides or skin, the trophies must:U.K.
(a)
have been either:
(ii)
dry- or wet-salted for a minimum of 14 days before dispatch, or
(iii)
[preserved by a treatment other than tanning approved by the Commission. Those measures, designed to amend non-essential elements of this Regulation, shall be adopted in accordance with the regulatory procedure with scrutiny referred to in Article 33(3);]
(b)
be packaged, immediately after treatment, without being in contact with other products of animal origin likely to contaminate them, in individual, transparent and closed packages so as to avoid any subsequent contamination; and
(c)
be accompanied by a document or certificate certifying that the above conditions have been met.
B.ImportationU.K.
5.Member States must authorise the importation of treated game trophies from birds and ungulates, being solely bones, horns, hooves, claws, antlers, teeth, hides or skins, from third countries if:U.K.
(a)
a certificate that conforms to the model laid down in Chapter 6(A) of Annex X accompanies them; and
(b)
they comply with the requirements of paragraphs 3 and 4. However, in the case of dry-salted or wet-salted skins transported by ship, the skins need not be salted 14 days before dispatch, provided that they are salted for 14 days before importation[;]
(c)
[they come from a third country appearing on the list set out in part XV (A) of Annex XI.]
6.Member States must, in accordance with the requirements of paragraph 7, authorise the importation of game trophies from birds and ungulates consisting of entire anatomical parts, not having been treated in any way, from third countries:U.K.
(a)
[that appear on the lists set out in part XV(B) and (C) of Annex XI as appropriate; and]
(b)
from which the importation of all categories of fresh meat of the corresponding species is authorised.
7.Member States must authorise importation of the game trophies referred to in paragraph 6 if:U.K.
(a)
they come from animals originating in an area not subject to restrictions as a result of the presence of serious transmissible diseases to which animals of the species concerned are susceptible;
(b)
they were packaged, immediately after treatment, without being in contact with other products of animal origin likely to contaminate them, in individual, transparent and closed packages so as to avoid any subsequent contamination; and
(c)
a certificate conforming to the model laid down in Chapter 6(B) of Annex X accompanies them.
CHAPTER VIIIU.K.Requirements for wool, hair, pig bristles, feathers and parts of feathers
A.Raw materialU.K.
[1.
(a)
Unprocessed wool, unprocessed hair, unprocessed pig bristles and unprocessed feathers and parts of feathers must have been obtained from animals referred to in Article 6(1)(c) or (k). They must be securely enclosed in packaging and dry. However, in the case of unprocessed feather and part of feathers sent directly from the slaughterhouse to the processing plant, the competent authority may allow derogation from the dry requirement, provided that:
(i)
all necessary measures are taken to avoid any possible spread of disease;
(ii)
the transport takes place in leak-proof containers and/or vehicles which must be cleansed and disinfected immediately after each use; and
(iii)
the Member State notifies the Commission when such derogation is given.
(b)
Movements of pig bristles from regions in which African swine fever is endemic are prohibited except for pig bristles that have:
(i)
been boiled, dyed or bleached; or
(ii)
undergone some other form of treatment which is certain to kill pathogenic agents, provided that evidence to this effect is submitted in the form of a certificate from the veterinarian responsible for the place of origin. Factory washing may not be regarded as a form of treatment for the purposes of this provision.]
2.The provisions of paragraph 1 do not apply to decorative feathers or feathers:U.K.
(a)
carried by travellers for their private use; or
(b)
in the form of consignments sent to private individuals for non-industrial purposes.
B.ImportationU.K.
3.Member States must authorise the importation of pig bristles from third countries or, in case of regionalisation according to Community legislation, regions thereof, if:U.K.
(a)
the pig bristles were obtained from animals originating, and slaughtered in a slaughterhouse, in the country of origin; and
(b)
either:
(i)
where no case of African swine fever has occurred during the previous 12 months, a certificate conforming to the model laid down in Chapter 7(A) of Annex X accompanies the consignment; or
(ii)
where one or more cases of African swine fever have occurred during the previous 12 months, a certificate conforming to the model laid down in Chapter 7(B) of Annex X accompanies the consignment[;]
(c)
[they come from a third country that appears on the list of part VIII of Annex XI as appropriate.]
4. [Member States must authorise the importation of unprocessed wool and hair, if they are:] U.K.
(a)
securely enclosed in packaging and dry; and
(b)
sent directly to the technical plant or to an intermediate plant in conditions such that any spread of pathogenic agents is avoided.
[5. The importation of unprocessed feathers and parts of feathers is prohibited. U.K.
Member States must authorise the importation of processed feathers and parts of feathers if:
(a)
they are treated decorative feathers, treated feathers carried by travellers for their private use or consignments of treated feathers sent to private individuals for non-industrial purposes; or
(b)
they are accompanied by a commercial document stating that the feathers or parts of feathers have been treated with a steam current or by another method ensuring the inactivation of pathogens and are securely enclosed in packaging and dry.]
[CHAPTER IX U.K. Requirements for apiculture products
A. Raw material U.K.
[1. Apiculture by-products intended exclusively for use in apiculture must:] U.K.
(a)
not come from an area which is subject of a prohibition order associated with an occurrence of:
(i)
American foulbrood ( Paenibacillus larvae larvae ), except where the competent authority has assessed the risk to be negligible, issued a specific authorisation for use only in that Member State, and taken all other necessary measures to ensure no spread of that disease;
(ii)
acariosis ( Acarapis woodi (Rennie), except where the area of destination has obtained additional guarantees in accordance with Article 14(2) of Directive 92/65/EEC () ;
(iii)
small hive beetle ( Aethina tumida ); or
(iv)
Tropilaelaps spp. ( Tropilaelaps spp); and
(b)
meet the requirements provided for in Article 8(a) of Directive 92/65/EEC.
B. Importation U.K.
2. As the small hive beetle and Tropilaelaps spp. are not present in the Community, the following additional safeguards concerning importation of apiculture products have to be laid down. U.K.
[3. Member States must authorise the importation of apiculture by-products, other than beeswax in the form of honeycomb, intended for use in apiculture if they: U.K.
(a)
come from third countries that appear on the list in Part XII of Annex XI;
(b)
either:
(i)
have been subjected to a temperature of – 12 °C or lower for at least 24 hours; or
(ii)
in the case of wax, the material has been refined or rendered before importation; and
(c)
are accompanied by a health certificate that conforms to the model set out in Chapter 13 of Annex X.]
[4. Member States must authorise the importation of beeswax for technical purposes, other than beeswax in the form of honeycomb, if it: U.K.
(a)
has been refined or rendered before importation; and
(b)
is accompanied by a commercial document attesting that refinement or rendering.
5. The importation of beeswax in the form of honeycomb shall be prohibited.] ] U.K.
CHAPTER XU.K.Requirements for bones and bone products (excluding bone meal), horns and horn products (excluding horn meal) and hooves and hoof products (excluding hoof meal) intended for use other than as feed material, organic fertilizers or soil improvers
1.Member States must authorise the importation of bones and bone products (excluding bone meal), horns and horn products (excluding horn meal) and hooves and hoof products (excluding hoof meal) to produce technical products if:U.K.
(a)
the products are dried before export and not chilled or frozen;
(b)
the products are conveyed only by land and sea from their country of origin direct to a border inspection post in the Community and are not transhipped at any port or place outside the Community;
(c)
following the document checks provided for in Directive 97/78/EC, the products are conveyed directly to the technical plant[;]
(d)
[they come from a third country appearing on the list set out in part XVII of Annex XI.]
2.Each consignment must be accompanied by:U.K.
(a)
a commercial document stamped by the competent authority supervising the establishment of origin, including the following information:
(i)
the country of origin,
(ii)
the name of the establishment of production,
(iii)
the nature of the product (dried bone/dried bone product/dried horns/dried horn products/dried hooves/dried hoof products), and
(iv)
the fact that the product was:
derived from healthy animals slaughtered in a slaughterhouse, or
dried for 42 days at an average temperature of at least 20 oC, or
heated for one hour to at least 80 oC to the core before drying, or
[ashed for one hour to at least 800 o C to the core before drying, or]
underwent an acidification process such that the pH was maintained at less than 6 to the core for at least one hour before drying, and
is not intended at any stage to be diverted for any use in food, feed material, organic fertilizers or soil improvers; and
(b)
[a declaration of the importer that conforms to the model laid down in Chapter 16 of Annex X and that must be in at least one official language of the Member State through which the consignment first enters the Community and in at least one official language of the Member State of destination.]
3.On dispatch to the Community territory, the material must be enclosed in sealed containers or vehicles or carried in bulk in a ship. If transported in containers, the containers, and in all cases all the accompanying documents, must bear the name and the address of the technical plant.U.K.
[4. Following the border check provided for in Directive 97/78/EC, and in accordance with the conditions laid down in Article 8(4) of that Directive, the material must be transported direct to the technical plant.] U.K.
5.Records must be kept of the quantity and nature of the material, during manufacture, in such a way as to ensure that the material has actually been used for the intended purposes.U.K.
[CHAPTER XI U.K. [Requirements for animal by-products for the manufacture of feed including petfood, and of technical products, excluding intermediate products as referred to in Article 1 of Commission Regulation (EC) No 2007/2006]
Member States must authorise the importation of animal by-products intended for the manufacture of feed including petfood, and for pharmaceutical products and other technical products if they:
1.
come from third countries appearing on the lists set out in part VI and VII(A) and (B) of Annex XI as appropriate;
2.
consist only of animal by-products referred to in Article 6(1)(a) to (j) and/or, when intended to be used for petfood, material derived from animals treated as referred to in the second paragraph of Article 28;
[however, animal by-products for use in feed for farmed fur animals or for use in raw petfood must consist of animal by-products referred to in Article 6(1)(a) and (b) only;]
3.
have been deep-frozen at the plant of origin or have been preserved in accordance with Community legislation in such a way to prevent spoiling between dispatch and delivery to the plant of destination;
4.
have undergone all precautions to avoid contamination with pathogenic agents;
5.
were packed in new packaging preventing any leakage;
6.
[are accompanied by a certificate that conforms to one of the models set out in Chapter 3(D), 3(F) or 8 of Annex X;]
7.
following the border checks provided for in Directive 97/78/EC, and in accordance with the conditions laid down in Article 8(4) of that Directive, they are transported directly either:
(a)
to a petfood or technical plant, which has given the guarantee that the animal by-products shall be used only for the purpose of producing petfood or technical products as appropriate, as specified by the competent authority if necessary, and shall not leave the plant untreated other than for direct disposal; or
(b)
to an intermediate plant; or
(c)
to an authorised and registered user or collection centre, which has given the guarantee that the animal by-products shall be used only for permitted purposes, as specified by the Competent Authority if necessary;
and
8.1..
in the case of raw material for petfood production derived from animals which have been treated with certain substances prohibited in accordance with Directive 96/22/EC, as referred to in the second paragraph of Article 28 of this Regulation, it shall:
(a)
be marked in the third country before entry into the territory of the Community by a cross of liquefied charcoal or activated carbon, on each outer side of each frozen block, in such a way that the marking covers at least 70 % of the diagonal length of the side of the frozen block and is at least 10 cm in width;
(b)
in the case of material which is not frozen, be marked in the third country before entry into the territory of the Community by spraying it with liquefied charcoal or by applying charcoal powder in such a way that the charcoal is clearly visible on the material;
(c)
be transported directly to:
(i)
the petfood plant of destination in accordance with point 7(a) above;
or
(ii)
an intermediate plant in accordance with point 7(b) above and from there directly to the petfood plant referred to under (i), provided that the intermediate plant:
only handles material covered by this point 8.1, or
only handles material destined for a petfood plant as referred to under (i);
and
(d)
be manipulated to remove the marking provided for in (a) and (b) only in the petfood plant of destination and only immediately prior to use of the material for the manufacture of petfood;
8.2..
where a consignment is made up of raw material, which has been treated as referred to in 8.1 above and other non-treated raw material, all the raw materials in the consignment must be marked as laid down in point 8.1(a) and (b) above.
8.3..
the marking provided for in point 8.1(a) and (b) and 8.2 shall remain visible from the dispatch and until the delivery to the petfood plant of destination.]
[CHAPTER XII U.K. Rendered fats from category 2 materials for oleochemical purposes
A. Processing standards U.K.
1. Rendered fats derived from category 2 material for oleochemical purposes must be produced using methods 1 to 5 as referred to in Annex V, Chapter III. U.K.
2. Rendered fats derived from ruminant animals must be purified in such a way that the maximum level of remaining total insoluble impurities does not exceed 0,15 % in weight; U.K.
B. Importation of rendered fats U.K.
3. Member States must authorise the importation of rendered fats derived from category 2 materials, intended to be processed using a method that at least meets the standards of one of the processes described in Annex VI, Chapter III, if it: U.K.
(a)
comes from a third country that appears on a Community list set out in part IV of Annex XI;
(b)
has been produced in accordance with this Regulation; and
(c)
is accompanied by a health certificate that conforms to the model set out in Chapter 10(B) of Annex X.
4. The rendered fats must be conveyed by land and/or sea from the country of origin direct to a border inspection post in the Community. U.K.
5. Following the checks provided for in Directive 97/78/EC, and in accordance with the conditions laid down in Article 8(4) of that Directive, the rendered fats must be conveyed to a category 2 oleochemical plant where they are to be processed into fat derivatives. U.K.
6. The health certificate referred to in paragraph 3 must state that: U.K.
(i)
the rendered fats will not be diverted for any use other than further processing by a method that at least meets the standards of one of the processes referred to in Chapter III of Annex VI; and
(ii)
the resulting fat derivatives shall only be used in organic fertiliser or soil improvers or other technical uses, other than in cosmetics, pharmaceuticals and medical devices.
7. The health certificate provided for in paragraph 3 must be presented to the competent authority at the border inspection post at the first point of entry of the goods into the Community, and thereafter a copy must accompany the consignment until their arrival at the plant of destination. U.K.
8. Following the checks provided for in Directive 97/78/EC, and in accordance with the conditions laid down in Article 8(4) of that Directive, the rendered fats shall be transported directly to the plant of destination.] U.K.
[CHAPTER XIII U.K. Fat derivatives
A. Processing standards U.K.
1. If rendered fat produced from category 2 material is used for the production of fat derivatives a method that at least meets the standards of one of the processes referred to in Chapter III of Annex VI shall be used. U.K.
B. Importation U.K.
2. Member States shall authorise the importation of fat derivatives only if a health certificate that conforms to the model set out in Chapters 14(A) or 14(B) of Annex X accompanies each consignment. U.K.
3. The health certificate referred to in paragraph 2 must state: U.K.
(a)
whether or not the fat derivatives derive from category 2 or 3 materials;
(b)
in the case of fat derivatives produced from category 2 material, that the products:
(i)
have been produced using a method that at least meets the standards of one of the processes referred to in Chapter III of Annex VI; and
(ii)
shall only be used in organic fertiliser or soil improvers or other technical uses, other than in cosmetics, pharmaceuticals and medical devices.
4. The health certificate provided for in paragraph 2 must be presented to the competent authority at the border inspection post at the first point of entry of the goods into the Community, and thereafter a copy must accompany the consignment until its arrival at the plant of destination. U.K.
5. Following the checks provided for in Directive 97/78/EC, and in accordance with the conditions laid down in Article 8(4) of that Directive, the fat derivatives shall be transported directly to the plants of destination. U.K.
CHAPTER XIV U.K. Specific requirements for flavouring innards for the manufacture of pet food
The following conditions apply in addition to the requirements for approval laid down in Chapter I.
A. Raw Material U.K.
1. Only animal by-products referred to in Article 6(1)(a) to (j) may be used for the production of liquid/dehydrated processed products of animal origin used to enhance the palatability values of pet food. U.K.
B. Processing standards U.K.
2. Flavouring innards must have been submitted to a treatment method and parameters, which ensure that the product complies with the microbiological standards laid down in Annex VIII, paragraph 6 of Chapter II. After treatment, every precaution must be taken to ensure that the product is not exposed to contamination. U.K.
3. The end product must: U.K.
(a)
be packed in new or sterilised packaging; or
(b)
transported in bulk in containers or other means of transport that were thoroughly cleaned and disinfected with a disinfectant approved by the competent authority before use.
C. Importation U.K.
4. Member States must authorise the importation of flavouring innards if they: U.K.
(a)
come from third countries that appear on the list set out in part VII(C) of Annex XI;
(b)
come from petfood plants approved by the competent authority of the third country meeting the specific conditions laid down in Article 18;
(c)
have been produced in accordance with this Regulation; and
(d)
are accompanied by a health certificate that conforms to the model set out in Chapter 3(E) of Annex X.]
[CHAPTER XV U.K. Requirements for horns and horn products, excluding horn meal, and hooves and hoof products, excluding hoof meal, intended for the production of organic fertilizers or soil improvers
A. Placing on the market U.K.
The placing on the market of horns and horn products, excluding horn meal, and hooves and hoof products, excluding hoof meal, intended for the production of organic fertilizers or soil improvers shall be subject to the following conditions:
1.
they must originate from animals that:
(a)
either have been slaughtered in a slaughterhouse, after undergoing an ante-mortem inspection, and were found fit, as a result of such inspection, for slaughter for human consumption in accordance with Union legislation; or
(b)
did not show clinical signs of any disease communicable through that product to humans or animals.
2.
they must have undergone a heat treatment for one hour at a core temperature of at least 80° C;
3.
the horns must have been removed without opening the cranial cavity;
4.
at any stage of processing, storage or transport, every precaution shall have been taken to avoid cross-contamination;
5.
they shall be packed either in new packaging or containers; or transported in vehicles or bulk containers which have been disinfected prior to loading using a product approved by the competent authority;
6.
the packaging or containers must:
(a)
indicate the type of product (horns, horn products, hooves or hoof products);
(b)
be clearly labelled ‘ NOT FOR HUMAN AND ANIMAL CONSUMPTION ’ ;
(c)
be marked with the name and address of the approved technical or storage plant of destination.
B. Importation U.K.
Member States shall authorise the importation of horns and horn products, excluding horn meal, and hooves and hoof products, excluding hoof meal, intended for the production of organic fertilizers or soil improvers provided that they:
1.
come from a third country appearing on the list referred to in Part XVIII of Annex XI;
2.
have been produced in accordance with point A of this Chapter;
3.
are accompanied by a health certificate that conforms to the model set out in Chapter 18 of Annex X, duly completed and signed by the official veterinarian;
4.
are conveyed following the veterinary checks in the border inspection post at the point of entry into the Union provided for in Directive 97/78/EC, and in accordance with the conditions laid down in Article 8(4) of that Directive, directly to an approved technical plant or an approved storage plant.]
ANNEX IXU.K.RULES APPLICABLE TO THE USE OF CERTAIN CATEGORY 2 AND CATEGORY 3 MATERIAL FOR THE FEEDING OF CERTAIN ANIMALS IN ACCORDANCE WITH ARTICLE 23(2)
1.This Annex applies only to users and collection centres authorised and registered pursuant to Article 23(2)(c)(iv), (vi) and (vii). For the purposes of this Annex, ‘relevant material’ means the animal by-products specified in Article 23(2)(b) and products derived therefrom.U.K.
2.Relevant material must be transported to the users or to collection centres in accordance with Annex II.U.K.
[2a. Entire bodies of dead animals shall be handled as Category 2 material during collection and transportation, without prejudice to the requirement to remove the specific risk material for subsequent disposal before the rest of the body may be used for feeding as provided for in Article 23.] U.K.
3.Collection centres must:U.K.
(a)
comply at least with the following requirements of Annex V:
(i)
Chapter I, paragraphs 1(a), (b), (c), (d) and (f), 2, 3 and 4, and
(ii)
Chapter II, paragraphs 1, 2, 4, 5 and 9; and
(b)
have adequate facilities for destroying unused unprocessed relevant material, or send it to a processing plant or to an incineration or co-incineration plant in accordance with this Regulation.
Member States may authorise the use of a Category 2 processing plant as a collection centre.
4.In addition to the records required in accordance with Annex II, the following records must be kept in relation to relevant material:U.K.
(a)
in the case of final users, the quantity used and the date of use; and
(b)
in the case of collection centres:
(i)
the quantity treated in accordance with paragraph 5;
(ii)
the name and address of each final user buying the material;
(iii)
the premises to which the material is taken for use;
(iv)
the quantity dispatched; and
(v)
the date on which the material was dispatched.
5.Operators of collection centres supplying relevant material other than fish offal to final users, must ensure that:U.K.
(a)
it undergoes one of the following treatments (either in the collection centre or in a slaughterhouse approved by the competent authority in accordance with Community legislation):
(i)
denaturing with a solution of a colouring agent approved by the competent authority. The solution must be of such a strength that the colouring on the stained material is clearly visible, and the whole surface of all pieces of material have been covered with a solution as aforesaid either by immersing the material in, or spraying or otherwise applying the solution;
(ii)
sterilisation, that is to say boiling or steaming under pressure until every piece of material is cooked throughout; or
(iii)
any other treatment approved by the competent authority; and
(b)
it is packaged after treatment and before distribution in packaging that is clearly and legibly marked with the name and the address of the collection centre and the indication ‘not for human consumption’.
[ANNEX X U.K. MODEL HEALTH CERTIFICATES FOR THE IMPORTATION FROM THIRD COUNTRIES AND FOR THE TRANSIT THROUGH THE EUROPEAN COMMUNITY OF CERTAIN ANIMAL BY-PRODUCTS AND PRODUCTS DERIVED THEREFROM
Notes U.K.
(a) Veterinary certificates shall be produced by the exporting country, based on the models appearing in this Annex X, according to the layout of the model that corresponds to the animal by-products concerned. They shall contain, in the numbered order that appears in the model, the attestations that are required for any third country and, as the case may be, those supplementary guarantees that are required for the exporting third country or part thereof. U.K.
(b) The original of each certificate shall consist of a single page, both sides, or, where more text is required, it shall be in such a form that all pages needed are part of an integrated whole and indivisible. U.K.
(c) It shall be drawn up in at least one of the official languages of the EU Member State in which the inspection at the border post shall be carried out and of the EU Member State of destination. However, these Member States may allow other languages, accompanied, if necessary, by an official translation. U.K.
(d) If for reasons of identification of the items of the consignment, additional pages are attached to the certificate, these pages shall also be considered as forming part of the original of the certificate by the application of the signature and stamp of the certifying official veterinarian, in each of the pages. U.K.
(e) When the certificate, including additional schedules referred to in d), comprises more than one page, each page shall be numbered — ( page number ) of ( total number of pages ) — on its bottom and shall bear the code number of the certificate that has been designated by the competent authority on its top. U.K.
(f) The original of the certificate must be completed and signed by an official veterinarian. In doing so, the competent authorities of the exporting country shall ensure that the principles of certification equivalent to those laid down in Council Directive 96/93/EC are followed. U.K.
(g) The colour of the signature shall be different to that of the printing. The same rule applies to stamps other than those embossed or watermark. U.K.
(h) The original of the certificate must accompany the consignment at the EU border inspection post. U.K.
(i) If health certificates are used for consignments in transit, box No I.5 (Consignee) of the relevant health certificate shall be completed with the name and address of the border inspection post through which the consignment is intended to leave the European Community. U.K.
[CHAPTER 2 U.K.
Health certificate For milk, milk products, colostrum and colostrum products not intended for human consumption for dispatch to or transit through () the European Union] U.K.




[CHAPTER 4 (A) U.K. Health certificate For the import of blood and blood products from equidae for technical purposes, intended for dispatch to or for transit through ( 2 ) the European Union]
[CHAPTER 4(C)] U.K.
[CHAPTER 4 (D) U.K. Health certificate For treated blood products, excluding those of equidae, for the manufacture of technical products, intended for dispatch to or for transit through ( 2 ) the European Union]
[CHAPTER 18 U.K. Health certificate For horns and horn products, excluding horn meal, and hooves and hoof products, excluding hoof meal, intended for the production of organic fertilizers or soil improvers intended for dispatch to or for transit through ( 2 ) the European Union] ]
[ANNEX XI U.K. Lists of third countries from which Member States may authorise imports of animal by-products not intended for human consumption
The inclusion of a third country on one of the following lists is a necessary, but not sufficient, condition for the importation of the relevant products from that third country. Imports must also fulfil the relevant animal health and public health requirements. The following descriptions refer to the territories or parts thereof from which imports of certain animal by-products are permitted, as stated in the relevant animal health certificate or declaration laid down in Annex X.
[PART I U.K. List of third countries from which Member States may authorise imports of milk, milk products, colostrum and colostrum products (health certificate Chapter 2)
A.
Milk and milk products:
Third countries listed as authorised in any of the columns of Annex I to Decision 2004/438/EC.
B.
Colostrum and colostrum products:
Third countries listed as authorised in column ‘ A ’ of Annex I to Decision 2004/438/EC.]
PART II U.K. List of third countries from which Member States may authorise imports of processed animal proteins (excluding fishmeal) (health certificate, Chapter 1)
Third countries listed in Part 1 of Annex II to Commission Regulation (EU) No 206/2010 () .
PART III U.K. List of third countries from which Member States may authorise imports of fishmeal and fish oil (health certificate, Chapters 1 and 9)
Third countries listed in Annex II to Commission Decision 2006/766/EC () .
PART IV U.K. List of third countries from which Member States may authorise imports of rendered fats (excluding fish oil) (health certificate, Chapters 10(A) and 10(B))
Third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010.
PART V U.K. List of third countries from which Member States may authorise imports of blood products for feed material (health certificate, Chapter 4(B))
A. Blood products from ungulates U.K.
Third countries or parts of third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010, from which imports of all categories of fresh meat of the respective species are authorised.
B. Blood products from other species U.K.
Third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010.
PART VI U.K. List of third countries from which Member States may authorise imports of animal by-products and blood products (with the exception of those of equidae) intended for technical purposes including pharmaceuticals (health certificate, Chapters 4(C) and 8)
A. Blood products: U.K.
1.
Untreated blood products of ungulates:
Third countries or parts of third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010 from which imports of fresh meat of any domestic ungulate species is authorised and only for the period indicated in columns 7 and 8 of that Part,
2.
Untreated blood products of poultry and other avian species:
Third countries or parts of third countries listed in Part 1 of Annex I to Commission Regulation (EC) No 798/2008 () ,
3.
Untreated blood products of other animals:
Third countries listed either in Part 1 of Annex II to Regulation (EU) No 206/2010, in Part 1 of Annex I to Regulation (EC) No 798/2008, or in Part 1 of Annex I to Commission Regulation (EC) No 119/2009 () ,
4.
Treated blood products of any species:
Third countries listed in Part 1 to Annex II of Regulation (EU) No 206/2010, in Part 1 of Annex I to Regulation (EC) No 798/2008, or in Part 1 of Annex I to Regulation (EC) No 119/2009,
B. Animal by-products for pharmaceutical use: U.K.
Third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010, in Part 1 of Annex I to Regulation (EC) No 798/2008, or in Part 1 of Annex I to Regulation (EC) No 119/2009 and the following third countries:
(JP) Japan,
(PH) Philippines,
(TW) Taiwan.
C. Animal by-products for technical purposes other than pharmaceutical uses: third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010 from which imports of that category of fresh meat of the respective species is authorised, in Part 1 of Annex I to Regulation (EC) No 798/2008, or in Part 1 of Annex I to Regulation (EC) No 119/2009. U.K.
PART VII(A) U.K. List of third countries from which Member States may authorise imports of animal by-products for the manufacture of petfood (health certificate, Chapter 3(F))
A. Animal by-products from equidae and animals of the bovine, ovine, caprine, and porcine species, including farmed and wild animals: U.K.
Third countries or parts of third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010, from which imports of fresh meat for human consumption of those species of animals is authorised.
B. Raw material from poultry including ratites and wild game-birds: U.K.
Third countries or parts of third countries from which Member States authorise imports of fresh poultrymeat, which are listed in Part 1 of Annex I to Regulation (EC) No 798/2008.
C. Raw material from fish: U.K.
Third countries listed in Annex II to Decision 2006/766/EC.
D. Raw material from other wild land mammals and Leporidae. U.K.
Third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010 or in Part 1 of Annex I to Regulation (EC) No 119/2009, from which Member States authorise imports of fresh meat from the same species.
PART VII(B) U.K. List of third countries from which Member States may authorise imports of raw petfood intended for dispatch to the European Union for direct sale or animal by-products to be fed to farmed fur animals (health certificate, Chapter 3(D))
Third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010 or in Part 1 of Annex I to Regulation (EC) No 798/2008, from which Member States authorise imports of fresh meat from the same species and where only bone in meat is authorised.
In the case of fish materials, third countries listed in Annex II to Decision 2006/766/EC.
PART VII(C) U.K. List of third countries from which Member States may authorise imports of flavouring innards for use in the manufacture of petfood, intended for dispatch to the European Union (health certificate, Chapter 3(E))
Third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010 or in Part 1 of Annex I to Regulation (EC) No 798/2008, from which Member States authorise imports of fresh meat from the same species and where only bone in meat is authorised.
In the case of flavouring innards from fish materials, third countries listed in Annex II to Decision 2006/766/EC.
PART VIII U.K. List of third countries from which Member States may authorise imports of pig bristles (health certificate, Chapter 7(A) and 7(B))
A. For untreated pig bristles, third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010, which are free of African swine fever for the 12 months prior to the date of importation. U.K.
B. For treated pig bristles, third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010, which may not be free of African swine fever for the 12 months prior to the date of importation. U.K.
PART IX U.K. List of third countries from which Member States may authorise imports of processed manure and processed manure products for the treatment of soil (health certificate, Chapter 17)
For processed manure and processed manure products, third countries listed in:
(a)
Part 1 of Annex II to Regulation (EU) No 206/2010;
(b)
Annex I to Commission Decision 2004/211/EC () ; or
(c)
Part 1 of Annex I to Regulation (EC) No 798/2008.
PART X U.K. List of third countries from which Member States may authorise imports of petfood and dogchews (health certificate, Chapters 3(A), 3(B) and 3(C))
Third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010, and the following third countries:
(JP) Japan
(EC) Ecuador ()
(LK) Sri Lanka ()
(TW) Taiwan ()
PART XI U.K. List of third countries from which Member States may authorise imports of gelatine, hydrolysed protein, collagen, dicalcium phosphate and tricalcium phosphate (health certificate, Chapters 11 and 12)
Third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010, and the following third countries:
(KR) South Korea ()
(MY) Malaysia ()
(PK) Pakistan ()
(TW) Taiwan () .
[PART XII U.K. List of third countries from which Member States may authorise imports of apiculture products (health certificate Chapter 13)
Third countries listed in part 1 of Annex II to Regulation (EU) No 206/2010, and the following country:
PART XIII U.K. List of third countries from which Member States may authorise imports of blood and blood products of equidae (health certificate, Chapter 4(A))
A. Untreated blood and blood products: Third countries or parts of third countries listed in Annex I to Decision 2004/211/EC, from which the importation of equidae for breeding and production is allowed. U.K.
B. Treated blood products: third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010, from which Member States authorise imports of fresh meat of domestic equidae. U.K.
PART XIV U.K. List of third countries from which Member States may authorise imports of hides and skins of ungulates (health certificate, Chapters 5(A), 5(B) and 5(C))
A. For fresh or chilled hides and skins of ungulates, third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010, from which Member States authorise imports of fresh meat from the same species. U.K.
B. For treated hides and skins of ungulates, third countries or parts of third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010. U.K.
C. For treated hides and skins of ruminants that are intended for dispatch to the Union and which have been kept separate for a period of 21 days or which are to undergo transport for a period of 21 uninterrupted days before importation, any third country. U.K.
PART XV U.K. List of third countries from which Member States may authorise imports of game trophies (health certificate, Chapters 6(A) and 6(B))
A. For treated game trophies of birds and ungulates, being solely bones, horns, hooves, claws, antlers, teeth, hides or skins, any third country. U.K.
B. For game trophies of birds consisting of entire parts not having been treated, third countries listed in Part 1 of Annex I to Regulation (EC) No 798/2008, from which Member States authorise imports of fresh poultry meat, and the following countries: U.K.
(GL) Greenland
(TN) Tunisia.
C. For game trophies of ungulates consisting of entire parts not having been treated, third countries listed in the appropriate columns for fresh meat of ungulates in Part 1 of Annex II to Regulation (EU) No 206/2010, including any restrictions laid down in the column for special remarks for fresh meat. U.K.
PART XVI U.K. List of third countries from which Member States may authorise imports of egg products not intended for human consumption that could be used as feed material (health certificate, Chapter 15)
Third countries listed in Part 1 of Annex II to Regulation (EU) No 206/2010, and third countries or parts of third countries from which Member States authorise imports of fresh poultry meat, which are listed in Part 1 of Annex I to Regulation (EC) No 798/2008.
PART XVII U.K. List of third countries from which Member States may authorise imports of bones and bone products (excluding bone meal), horns and horn products (excluding horn meal) and hooves and hoof products (excluding hoof meal) intended for use other than as feed material, organic fertilisers or soil improvers (declaration, Chapter 16)
Any third country.
PART XVIII U.K. List of third countries from which Member States may authorise imports of horns and horn products (excluding horn meal) and hooves and hoof products, (excluding hoof meal) intended for the production of organic fertilizers or soil improvers (health certificate, Chapter 18)
Any third country.]